Good morning.
Welcome to today's edition of The Armchair Analyst, a 5-minute daily update on the ASX life-sciences sector.
Last Friday, the US Government issued the first three Priority Review Vouchers to psychedelics companies.
Compass Pathways - psilocybin for Treatment-Resistant Depression
Usona Institute - psilocybin for Major Depressive Disorder
Otskua - MDMA for PTSD
Big news for the psychedelics industry. Bigger news for my second Armchair Pick, Emyria (ASX: EMD).
We could see a psychedelic therapy approved in the US within the next 12 months, but that doesn’t answer the question: who actually delivers it?
So, what is a Priority Review Voucher? And why are big pharma companies paying US$200M to get access to one?
Today, I'm doing a deep dive into the US Priority Review Voucher system.
How it works, what it's worth, and why the political maneuvering to secure one for free can be one of the most valuable plays in biotech.
But first…
The Pulse Check
Chimeric Therapeutics (ASX: CHM) reports 60% Response Rate (Complete or Complete with Incomplete Count Recovery) for its Phase 1B CORE-NK trial for frontline high-risk blood cancer. (CHM)
🪑 I’m a CHM shareholder, but I had to read this one twice to answer the question: “How good is this news?”
First, CHM has two cancer trials: the first, Colorectal Cancer, is its lead trial, and the second is an off-the-shelf cell therapy for Blood Cancer.
These results come from the second trial for blood cancer.
Previously, CHM reported a 57% clinical response from 4 of 7 patients.
The wording of the announcement is incredibly unclear.
Are these results from 60% of the 25 patients “enrolled” into the trial (which would mean 15 clinical responses); OR
There are 25 patients enrolled in the trial, and the results are from 60% of the patients who are dosed (without stating how many patients this represents).
If it is the former, then the results are very good. Continuing to see a 60% clinical response in larger patient populations is a big plus.
If it is the latter, I would want more clarity on how many patients are specifically affected before making a judgment.
Clinuvel Pharmaceuticals (ASX: CUV) confirms that the FDA has removed the requirement of a cardiac polerisation study for SCENESSE® postmarketing authorisation. (CUV)
PYC Therapeutics (ASX: PYC) secures Orphan Drug Designation from the EMA for its VP-001 asset. (PYC)
Alterity Therapeutics (ASX: ATH) received FDA feedback on Phase 3 plans for its Multiple System Atrophy treatment. Final FDA meeting before Phase 3 is on track for mid-2026. (ATH)
🪑 It was great to hear Lawrance Gozlan present the Alterity story last week at the Monsoon Twilight Event.
He was asked the question: You completed your Phase 2 study in January last year. Why has it taken so long for you to reach this end-of-Phase 2 meeting with the FDA?
While the Phase 2 data was published (and promising), the company had to go back and conduct earlier-stage toxicology work required by the FDA before it could move towards Phase 3.
I’ve seen this play out a number of times with companies advancing drugs through the US. They want to access the data as soon as possible but neglect the important lab work required by the FDA. This can set the company back months after a good result (and snuff out momentum in the story).
It's definitely a risk that I’m watching out for, particularly those companies preparing for a Phase 3 in the US.
It appears that Alterity is over that hump, but it did set them back.
Syntara Limited (ASX: SNT) requests a trading halt pending FDA review of its correspondence regarding the Phase 2b trial protocol for its lead asset, Amsulostat. (SNT)
🪑 Don’t often see a trading halt for just an FDA correspondence on a clinical trial protocol.
Interesting, watch this space.
Noxopharm (ASX: NOX) Sofra™ platform for inflammation delivers the first results from a partner under the Material Transfer Agreement. (NOX)
🪑 This data is a good first step to move towards a more formal partnership.
Biome Australia (ASX: BIO) files patent application for next-gen probiotic strain BMB18. Human trial set for Q4 FY26. (BIO)
Report: Scientists want evidence for AI in medicine (Second Opinion, Nature)
🪑 This article in Nature is giving off big “old man yells at cloud” vibes.
It is not unexpected that, as new technologies emerge, a conservative subset of the population will push back (particularly in the healthcare sector, where the conservative voice is loud).
However, I think AI has proven (and continues to prove), particularly with AI decision support frameworks for diagnostics (like in radiology and cardiology), the benefits.
The Report Card
Oneview Healthcare (ASX: ONE) reports a cash balance of A$17.1M as of 31 March 2026. Cash inflows of €3.2M. (ONE)
🪑 Not ONE’s strongest quarter for sales (down from the previous quarter, €3.5M)
You can tell how the company feels about their quarterly results by what they chose to focus on first. The introduction was all about the capital raise and shoring up the balance sheet, THEN the commercial updates on page 2.
HITIQ Limited (ASX: HIQ) reports March quarter sales of $294k, a 223% increase from $91k in the December quarter. (HIQ)
🪑 Some of the first meaningful sales out of HIQ for a while. Let’s see if they can back it up next quarter.
Cash Injection
Paradigm Biopharmaceuticals (ASX: PAR) completes an upsized $14M placement, exceeding its $8M target at $0.19 per share. (PAR)
🪑 Nice job.
Under the Microscope
Last Friday, the US Government issued the first three Priority Review Vouchers to psychedelics companies.
Compass Pathways - psilocybin for Treatment-Resistant Depression
Usona Institute - psilocybin for Major Depressive Disorder
Otskua - MDMA for PTSD

(Source, CNN Health)
This is a big step toward bringing psychedelic therapy to the US market. We could potentially see one of these three companies with an approved product inside 12-18 months.
It's also the first concrete outcome of the Trump administration's executive order last week to "fast-track" psychedelic medicine for mental health.
But while the drug might get to market much quicker, there's a looming question:
Who actually delivers the drug, and the therapy?
My Second Armchair Pick, Emyria (ASX: EMD), is very well placed to answer this question.
So it's clinical delivery, NOT drug development, that is emerging as a primary constraint to the rollout.
My full take 👉 My Second Armchair Pick: Emyria (ASX: EMD)
So, what exactly is a Priority Review Voucher? And why are big pharma paying US$200M to get one?
So, what are Priority Review Vouchers?
There are two types of Priority Review Vouchers:
Standard PRV - Transferable vouchers under existing FDA programs (neglected tropical diseases, rare pediatric diseases, bioweapon defences).
Commissioner’s PRV - A newer, nontransferable voucher granted at the FDA’s discretion to accelerate review for products aligned with national priorities (public health crises, breakthrough therapies, unmet need, supply chain resilience, affordability)
Think of a PRV as the "fast pass" at Movie World.
While the average punters queue up in the slow line, the fast pass gets you there, well… faster.

These fast passes are very valuable because in biotech land…
Time is money.
Bringing a drug to market a few months earlier can mean hundreds of millions in sales, particularly with tight exclusivity windows ticking down from approval.
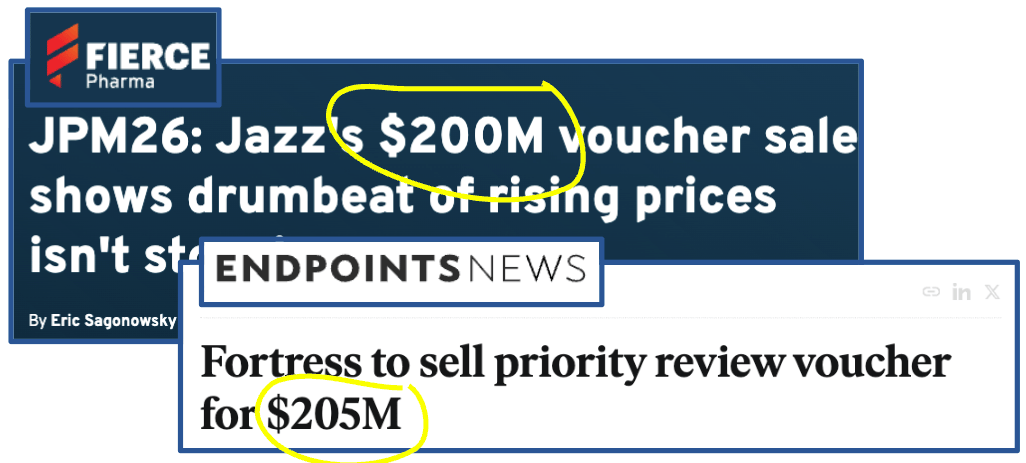
That's why a single PRV today changes hands for around US$200M.
Standard, Transferable Priority Review Vouchers
The first PRVs were established in 2007 as a "thank you" to companies developing treatments for rare tropical diseases.
Illnesses that were common and serious, yet receive far too little funding and attention.
The idea was simple: create a market-based incentive to attract drug developers to neglected areas.
The program was later extended to biodefence and rare pediatric diseases.
How does it work?
When a company gets FDA approval for a drug targeting one of these qualifying conditions, it's issued a voucher.
(Think of it as a thank-you for working on something important.)
The voucher buys faster FDA review for a future drug and, crucially, can be sold to another company.
Two PRVs have already traded this year for US$200M each.
Just last week, the FDA approved Regeneron's gene therapy product for an ultra-rare form of genetic hearing loss.
The approval came with a PRV attached, given the disease's rarity.
That's effectively a US$200M tradeable asset for Regeneron, on top of whatever the drug itself earns.
ASX-listed companies leveraged to Priority Review Vouchers
There are two ASX-listed companies that are specifically targeting the Priority Review Voucher scheme (but in very different areas).
Neuren Pharmaceuticals (ASX: NEU).
Neuren develops treatments for serious neurological disorders, particularly rare pediatric conditions.
Its first product, DAYBUE for Rett Syndrome, secured a PRV that sold for US$150M in 2024 (Neuren received US$50M under its licensing deal with Acadia).
Every indication Neuren goes after is a rare pediatric condition.
Meaning every new approval carries another potential PRV.
The Phase 3 program for Phelan-McDermid Syndrome has begun recruiting.
At today's PRV prices, that's a ~US$200M one-off payment stacked on top of the drug's future cash flows on approval.
Island Pharmaceuticals (ASX: ILA)
Island is developing a treatment for Marburg virus, a rare, deadly virus with a history of being developed as a bioweapon.
The PRV Island is chasing falls under the biodefence category.
The company has FDA approval to use the "Animal Rule" pathway, a little-known regulatory shortcut for treatments against viruses too deadly to test in humans (like anthrax and smallpox).
It means no Phase 2 or Phase 3 efficacy trial in humans is required.
Animal trials assess efficacy. A Phase 1 trial in humans assesses safety.
Island already has Phase 1 data (inherited from the company that sold them the drug) and just needs additional animal data to prove efficacy.
Again, add a US$200M one-off to the drug's future cash flows if it is approved.
A second fast pass lane opens the Commissioner’s National Priority Review Voucher
Last year, the Trump Administration announced a new Priority Review Voucher.
The Commissioner’s National Priority Voucher (CNPV)...
A pilot program designed to speed up drug approvals in specific therapies critical to the national priorities:
Public health emergencies;
Breakthrough innovations;
Major unmet medical needs;
Supply chain resilience; or
Affordability
In other words, a second “fast pass” line has opened up.

But unlike the Standard PRV, the Commissioner's voucher is the administration's political lever.
A tool to negotiate better drug-pricing terms and play kingmaker in industries it wants to shape.
That is what is happening right now in the psychedelic therapy space.
Three companies, each with a Commissioner’s Priority Review Voucher.
Importantly, these are not transferable and are specifically designed to get a drug across the line.
So, there are two ways to move into the fast lane.
Pay up, or play up.
Lessons for late-stage drug developers
Traditionally, every late-stage biotech has two strategic pillars heading into the end of Phase 3:
A clinical strategy - proving that the drug works.
A commercialisation strategy - getting the drug sold.
But there is a third pillar that is emerging now:
A political strategy - navigating the kingmaker dynamics of the current administration so your drug doesn't get stuck in the regulatory slow lane.
Which raises the more interesting question: does the new fast lane hurt or help the value of the traditional, saleable PRV?
My first instinct was that it hurts.
If there's a second, essentially free way to fast-track a drug, the traditional PRV should lose its scarcity premium.
But the opposite looks more likely.
Because with the second fast lane open, every company not on it gets pushed further back in the regular queue.
The cost of being stuck in slow traffic rises, making paying US$200M for a saleable PRV more attractive.
The market seems to agree.
Both sales this year cleared at US$200M, up from US$150M in 2024. Prices are rising, not falling.
The bottom line for late-stage biotech…
If you're not thinking about your political strategy, you may be in for a long slog, even if your clinical and commercial strategies are flawless.
See you all tomorrow,
The Armchair Analyst.