Under the Microscope
There is an old saying that I like…
Throw 3 balls in the air, and you’ll struggle to catch one.
But throw just one ball in the air, and I’ll look like Steve Smith.
Every time…
Now, Syntara (ASX: SNT) can feel like a 3-ball company…
(multiple assets, multiple shots on goal, each going after a different target)
But let me see if I can find a common narrative thread to tie it all together.
Challenge, accepted.
What’s the story?
At its core, Syntara (ASX: SNT) is a drug development company.
(duh!)
Its lead asset, Amsulostat, is for a rare bone marrow disease (Myelofibrosis).
It has Phase 2a that has a strong safety profile and competitive efficacy compared to the current standard of care.
Now, there is a drug on the market that treats Myelofibrosis.
A JAK inhibitor called ruxolitinib, it does US$1.9 billion in Myelofibrosis alone.
But here's the problem.
Up to 70% of patients have a poor response to it. The side effects are brutal. And once it stops working… there's basically nothing else.
Oh, and it's coming off patent. Europe in 2027. US in 2028.
So that leaves a gaping hole in the market for someone to come in with a better asset, a better solution.
That’s Syntara’s “Main Bet”.
Now, Syntara has two other assets (let’s call them Side bet 1 and Side bet 2) for two other distinct conditions.
Sidebet 1: A topical cream to remove skin scarring.
A big, big market, particularly for cosmetic surgery.
Sidebet 2: A neuroinflammatory drug for a sleep condition that is strongly linked with Parkinson’s Disease.
Funded by someone else.
There are a few other trials underway as well, but all are funded by grants or third parties.
… As I said, at its core, Syntara is a drug development company.
That’s what it does: it develops drugs.
Lots of them.
All R&D is done in-house, so the IP for each of its assets is owned by Syntara.
The goal with each asset is to eventually partner it off.
Now every biotech says that there is a huge market for their asset, and partners will line up once they see the data.
But very few actually sell to big pharma.
Not in Syntara’s case.
That Sidebet 2 drug I was talking about before?
Well, it was actually sold to the big pharma company Boehringer Ingelheim in 2015 for an upfront payment of A$39 million and A$625 million in development milestones.
But just before Phase 3 commenced, a last-minute toxicology study showed an unexpected enzyme that switched off in the brain.
It didn’t necessarily affect the safety results, but it was enough to scare off Boehringer Ingelheim.
And they sold it back to Syntara for nothing.
That enzyme, that switched off… turns out that researchers working on this sleep problem that caused Parkinson’s Disease were looking for this exact thing.
So back to the clinic it goes.
The CEO, Gary Phillips, confidently said, "I sold it once; I might just sell it again."
… As I said, at its core, Syntara is a drug development company.
But with a proven track record of selling assets to big pharma.
There is a flurry of data readouts coming soon for Syntara.
With Side Bet 1 (scaring) set for the second half of the year and Side Bet 2 (Parkinson’s Disease) this side of June.
This Mainbet/Sidebet strategy is widely used in the mining world.
Often, companies will hold multiple assets in multiple jurisdictions and work them up whenever the underlying commodity is in favour.
Biotech companies don’t have the luxury of an external commodity making a dormant project suddenly valuable.
(As I’ve said before, there is no “gold price” equivalent for heart attacks)
But, because much of the research has been funded by others ($11.5 million in grant funding in the last three years), these shots on goal are essentially free rolls for Syntara.
… As I said, at its core, Syntara is a drug development company.
But with other people paying to develop its assets.
This is the business model that makes Syntara’s “multiple shots on goal” strategy actually work.
Now, let’s take a look at what they’ve been able to develop…
Main Bet: Bone Marrow (Blood) Cancer
Bone marrow is like the body's factory.
Creating soldiers (blood cells) that fight off infection, repair damage, and keep the body alive.
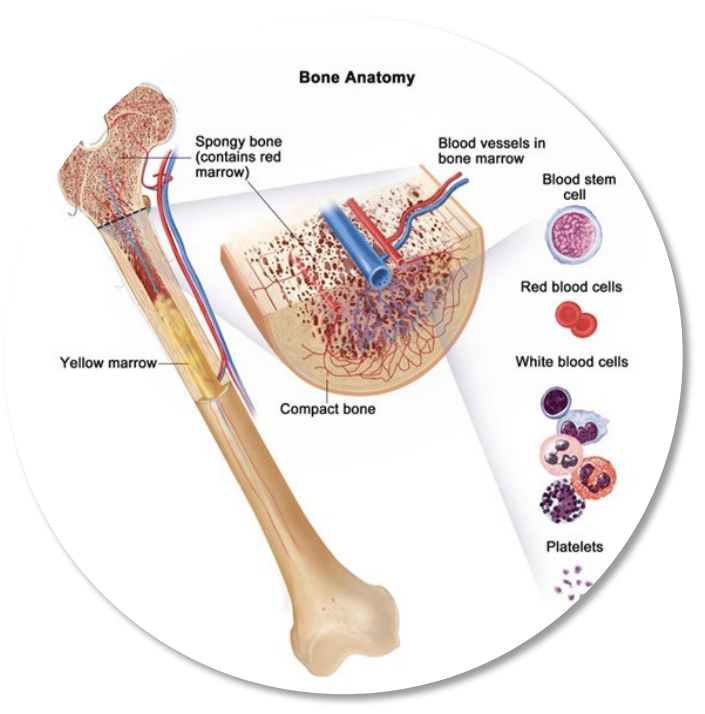
Myelofibrosis is a rare bone marrow cancer caused by a buildup of scar tissue (fibrosis), which reduces blood cell production.
The spleen tries to pick up the slack; it swells, and patients grow weaker.
Median survival from diagnosis: five years.
In the last three years, three myelofibrosis companies have been bought for between US$1.7 billion and US$2.9 billion each.
Novartis paid US$2.9 billion for a company just to get its hands on a rival drug. But a safety signal emerged, patients were showing higher rates of a life-threatening complication, and now that drug is delayed for years.
Novartis wrote down US$800 million from the deal.
(ouch!)
In December 2024, Syntara presented Phase 2a results at the world's largest blood cancer conference.
46% of patients had a major reduction in symptoms by week 12. By week 38, that was 80%.
No safety issues, with results getting better over time.
(Safety is super important with this one)
The problem was that the market expected Syntara to go directly to a Phase 3 study, but the FDA wanted more data in a Phase 2 before the pivotal study.
Even though the data was really good, the market hates delays, and so it was punished:

The stock has recovered a bit since then, but the market will be waiting for the results of FDA meeting, which will give guidance on the development plan.
(Expected mid-year)
Now, the FDA development plan itself won’t move the stock, but any outcome from the “partner engagement” discussion afterwards might.
Development plan unlocks partner engagement.
But even if nothing comes from partnering, Syntara still has the option to take the asset to the clinic and develop it through to the end of Phase 2.
Side Bet 1: Tissue Scaring (topical cream)
The global scar treatment market is valued at around US$30 billion annually.
Laser treatments are the fastest-growing part of it. People book in. They sit through multiple sessions. They spend thousands.
And at the end of it?
The scar looks a little better. A little less red. A little flatter.
But it's still there.
Laser doesn't fix the underlying structure of the scar, it just resurfaces on top of it.
Syntara’s product changes the underlying structure of the scar.
Which means it could potentially work on scars that are years, even decades, old.
(perfect for the cosmetics industry)
Now, Phase 1 showed the cream was working at a biological level, and biomarkers confirmed it was hitting its target. But three times a week wasn't enough to actually shift the scar.
So the trial has been reformulated to once a day at a higher dosage.
Designed to do what the first trial couldn’t do.
The trial design is clever too, patients are their own control, the same scare gets both the cream and the placebo.
You see the difference in real time, on the same person.
Results coming in the second half of this year.
Sidebet 2: Parkinson’s disease
As I mentioned before, this asset was sold to Boehringer Ingelheim in 2015 and then returned to Sytnara four years later.
It was discovered that an unexpected enzyme switched off in the brain…
Turns out that researchers working on this sleep problem that causes Parkinson’s Disease were looking for this exact thing.
So Syntara set up a new trial funded by Parkinson’s UK.
Results scheduled for this side of June.
Tying it all together
Alright, let’s see if I can catch all three balls at once here.
Syntara is a drug development company.
With the goal of incubating internally developed science (supported by grants and other funding) and with a track record of selling assets to big pharma.
That’s what makes Syntara a 1-ball company.
It’s an asset incubator, with a track record of getting others to pay for the development of its assets and selling to big pharma at the right time.
While there are multiple shots on goal, it is clear that the Main Bet (bone marrow cancer) is the hero.
It has the most data and the biggest unmet need.
BUT I wouldn’t discount either of its two other side bets; side bets can quickly become main bets with good data.
So, what’s the bet?
Short term: Syntara gets a good readout and re-rate on the results from either of its Side Bets… and you don’t have to wait long.
Long term: Syntara is able to sell off one or multiple assets (with the most valuable being its Bone Marrow Cancer treatment), and leverage its in-house R&D to come up with new pipeline assets to develop and sell.
A big thank you to Syntara CEO Gary Phillips for speaking to me on Monday and sharing the Syntara story.
See you all tomorrow,
The Armchair Analyst
The Pulse Check
Island Pharmaceuticals (ASX: ILA) signs a Cooperative Research and Development Agreement with two US military scientific research institutes: USAMRIID and The Geneva Foundation. (ILA)
🪑USAMRIID will consult on ILA's planned trial design for its Marbourg disease treatment; they have deep sector expertise (in fact, they ran the previous studies on ILA’s asset when the asset was owned by BioCryst in 2014).
The Geneva Foundation is effectively a peak body that brings ILA closer to the US Military.
Two very good partners for ILA’s bio-defence products.
The Supreme Court of Victoria has approved the merger scheme between Little Green Pharma (ASX: LGP) and Cannatrek. Shareholder vote set for April 10th. (LGP)
Control Bionics (ASX: CBL) secures a reimbursement code for its NeuroNode device in Germany. (CBL)
🪑 What this essentially means is that CBL can now negotiate with insurance providers in Germany to have its product reimbursed under the equivalent of Medicare/NDIS.
It’s no secret that the largest providers of assistive technology in Germany are Tobii, PRC Satillio and NextLevel - the three companies that CBL just signed a distribution agreement with.
Reimbursement essentially unlocks German (and Europe) for CBL, without needing to set up its own sales team at all.
AdAlta (ASX: 1AD) expands the clinical advisory board for its subsidiary AdCella, which has licensed a CAR-T cell therapy platform from China. (1AD)
🪑1AD is setting up the AdCella subsidiary to be the vehicle for licensing and developing clinical assets from China.
As a private entity, it will need to raise capital independently (around $20 million) to fund the scheduled clinical programs.
These board appointments move the company towards that funding, providing independent expertise that gives VC firms confidence to invest in private companies.
Rhythm Biosciences (ASX: RHY) signs a 12-month supply and distribution agreement with Digistain Limited to launch its geneType™ cancer diagnostic tests in the Philippines. (RHY)
🪑Good choice of partner, Digistain is a specialist international distributor and has multiple commercial networks across South Asia.
Cash Injection
PYC Therapeutics (ASX: PYC) completes retail entitlement offer, raising $47M at $1.50/share with a 40% take-up rate, following its $537M institutional raise earlier this year. (PYC)





