Good morning,
Welcome to today’s edition of The Armchair Analyst, a 5-minute daily update on the ASX life-sciences sector.
Another trading halt. Another heart-in-mouth moment.
Investors in the dark until the trading halt lifts.
This time, Lumos Diagnostics (ASX: LDX).
The upcoming FDA decision could be the difference between US$25 million and US$317 million in revenue over the next six years.
Lumos sells a finger-prick, point-of-care test to determine whether a respiratory infection is bacterial or not.
The US licensing deal signed last year with PHASE Scientific, worth US$317 million, was contingent on an FDA CLIA waiver.
Without it, the deal shrinks to 7% of the size, and just US$25 million over the next six years.
That's a US$300 million swing, in just one decision.
Big stakes, trading halt lifts tomorrow.
But first…
The Pulse Check
BCAL Diagnostics (ASX: BDX) expands the national rollout of its Avantect Pancreatic and Ovarian blood tests through Sonic Healthcare to Victoria, New South Wales and Queensland. (BDX)
🪑 It has been a quick ramp-up since the first Avantect test was launched in January. Will be interesting to see what uptake the company can get with more patient access to the tests.
Control Bionics (ASX: CBL) signs its first U.S. Letter of Intent for iOS-based speech devices with NextLevel projecting US$2.5-3.5M annual revenue. (CBL)
🪑 CBL signed a deal with NextLevel last month, and this is the first projected revenue under the deal.
Still, non-binding, but nice validation all the same (and NextLevel will pay US$100K upfront).
Rhythm Biosciences (ASX: RHY) completes the first commercial sale of its geneType™ cancer risk assessment test in the Philippines, marking its entry into Southeast Asia. (RHY)
Nexsen Limited (ASX: NXN) update on the global rollout of StrepSure® rapid point of care product for GBS. (NXN)
🪑 Meaty update this one, but here are the high-level takeaways:
It was confirmed that the FDA would need some US data from NXN for approvals (as I suspected). It will now move quickly to set up US sites and hospitals in collaboration with its CRO. FDA submission scheduled now for the end of the year.
The first markets NXN will target are Hong Kong, Malaysia, and India - keep an eye out for early commercialisation milestones, including partnerships with hospital networks.
Big manufacturing update - I’m less interested in this, but an important de-risking event.
AusSuper will increase insurance premiums by up to 40% due to a rise in Mental Health and Disability claims among younger generations. (AFR)
🪑 Interesting to see whether more insurance companies beyond Medibank will start supporting new psychedelic mental health therapies like those offered by Emyria (ASX: EMD).
Cash Injection
Osteopore (ASX: OSX) receives an upfront fee of $520,000 from Majeton as part of its distribution deal to commercialise its dental and maxillofacial implants in China, Hong Kong, and Macau. (OSX)
Gilgamesh Pharma Closes Oversubscribed US$60 Million Series A to Develop Next Generation Neuropsychiatric Therapies. (PR Newswire)
🪑 This was the first independent capital raised since AbbVie acquired Gilgamesh last year - the first and largest acquisition in the psychedelic therapy space.
M&A, Big Pharma Wants a Wife
Merck to acquire Terns Pharmaceuticals in oncology deal worth up to US$6.7B as it looks to solidify its oncology pipeline for a post-Keytruda world. (Reuters)
🪑 Massive sigh… this could have been Immutep.
Merck partners with Quotient Therapeutics on an Inflammatory Bowel Disease (IBD) drug discovery platform. US$20M upfront, US$2.2B in milestones. (Fierce Biotech)
🪑 Merk lit a fuse under the IBD indication with its $11 billion purchase of Prometheus Biosciences in 2023.
Will be interesting to see how this deal improves the fortune of ASX-listed Microba (ASX: MAP), which is looking for a pharma partner to take its IBD product through to a Phase 2.
Under the Microscope
Lumos Diagnostics (ASX: LDX) sells a finger-prick test to determine whether a respiratory infection is bacterial or viral.
A rapid, accurate evaluation to determine whether antibiotics are needed or not.
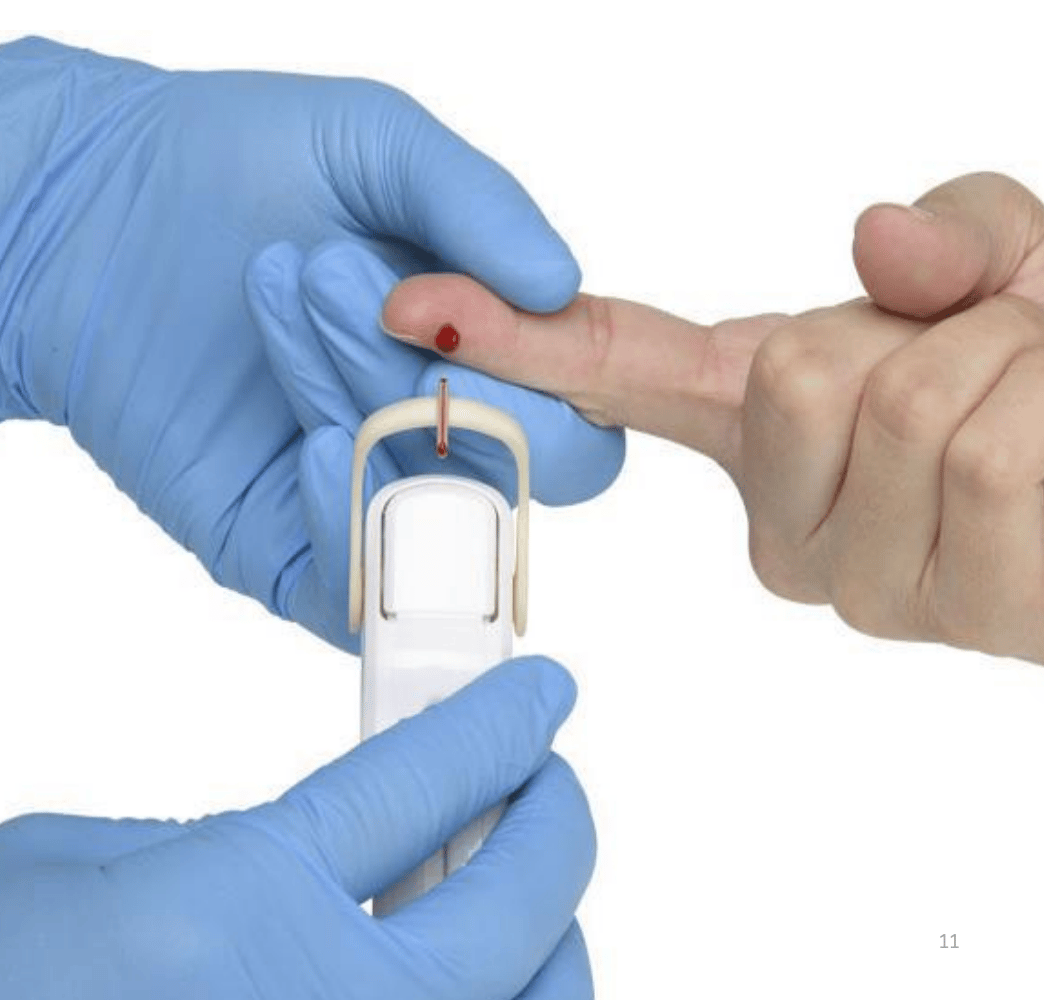
Before the market opened yesterday, Lumos entered a trading halt to provide an update on the FDA's response to the FebriDx CLIA Waiver Application.
(FebriDx is the name of its diagnostics product)

(Source, LDX Announcement)
A CLIA waiver allows a test to be used by a broad range of physicians.
Think urine pregnancy tests, rapid strep test, COVID-19 RAT tests.
WITHOUT the CLIA waiver, this product can only be sold in areas with high complexity testing.
Hospital labs, reference labs, etc…
WITH the CLIA waiver, the test can be used in standard physician offices and urgent care clinics.
The addressable market improves about 15-fold.
There is a lot of money at stake here too.
Last year in June, Lumos signed an exclusive distribution deal with PHASE Scientific to sell and distribute its FebriDx in the USA.
With the conditions of the deal heavily dependent on the outcome of this CLIA waiver…
If positive for Lumos, it will:
Get US$5M immediately up front.
Unlock the ability to sell its products to doctors and pharmacies (expanding its total addressable market from $66 million to $1 billion).
Unlock minimum order quantities to “reach up to US$317M (A$487M) over the life of the agreement” (six years).
If negative for Lumos,
No US$5M
Minimum order quantities will be set at 7%, with an expected contract value of US$25M (A$38M) over the six-year term.
This would be very bad.
Now, the minimum order quantities that make up the US$317M under the contract are just that…
Minimum quantities expected.
The company left enough wiggle room in the contract to adjust the commercial terms if they are not achieved.
So I see these revenue projections as more than aspirational but less than definitive.
The product will still need to sell.
Lumos has secured Medicare reimbursement for the product at around ~US$41.
Interestingly, another company listed on the ASX has its fate heavily tied to this deal as well.
Atomo Diagnostics (ASX: AT1).
On no news, the company was up 14% yesterday, with $211,000 of stock traded:
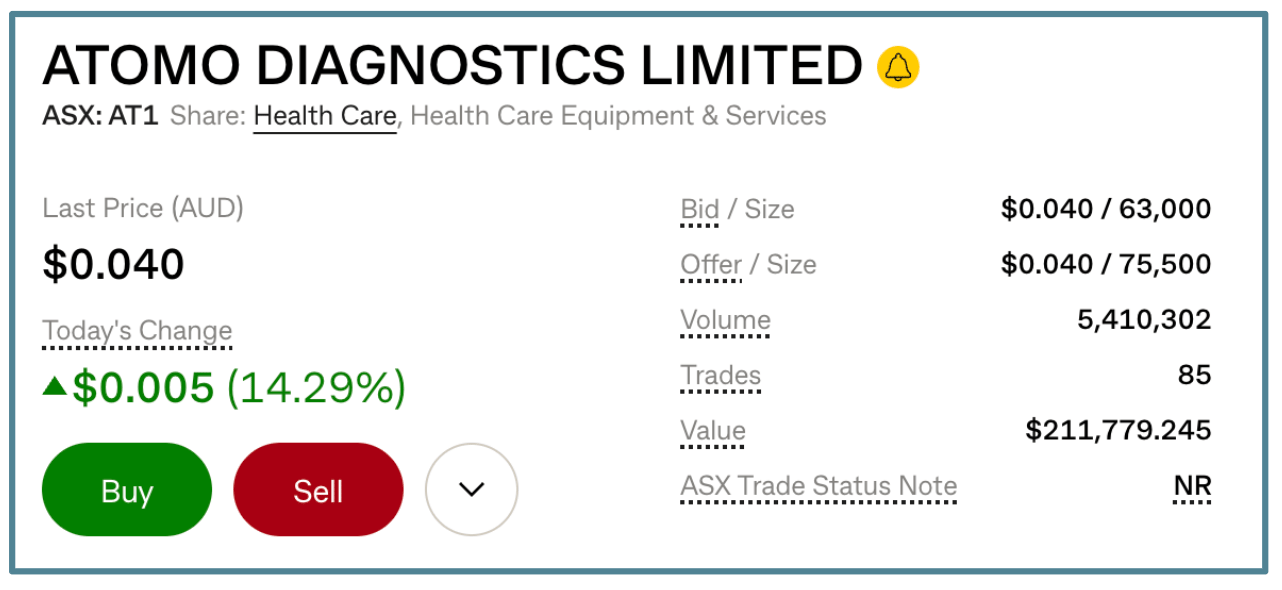
By 2:00 pm, the company was in a trading halt, with a “please explain” from the ASX released after the market close.
You see, Atomo sells the finger-prick shells to Lumos Diagnostics, which holds the IP for the lateral flow test inside.
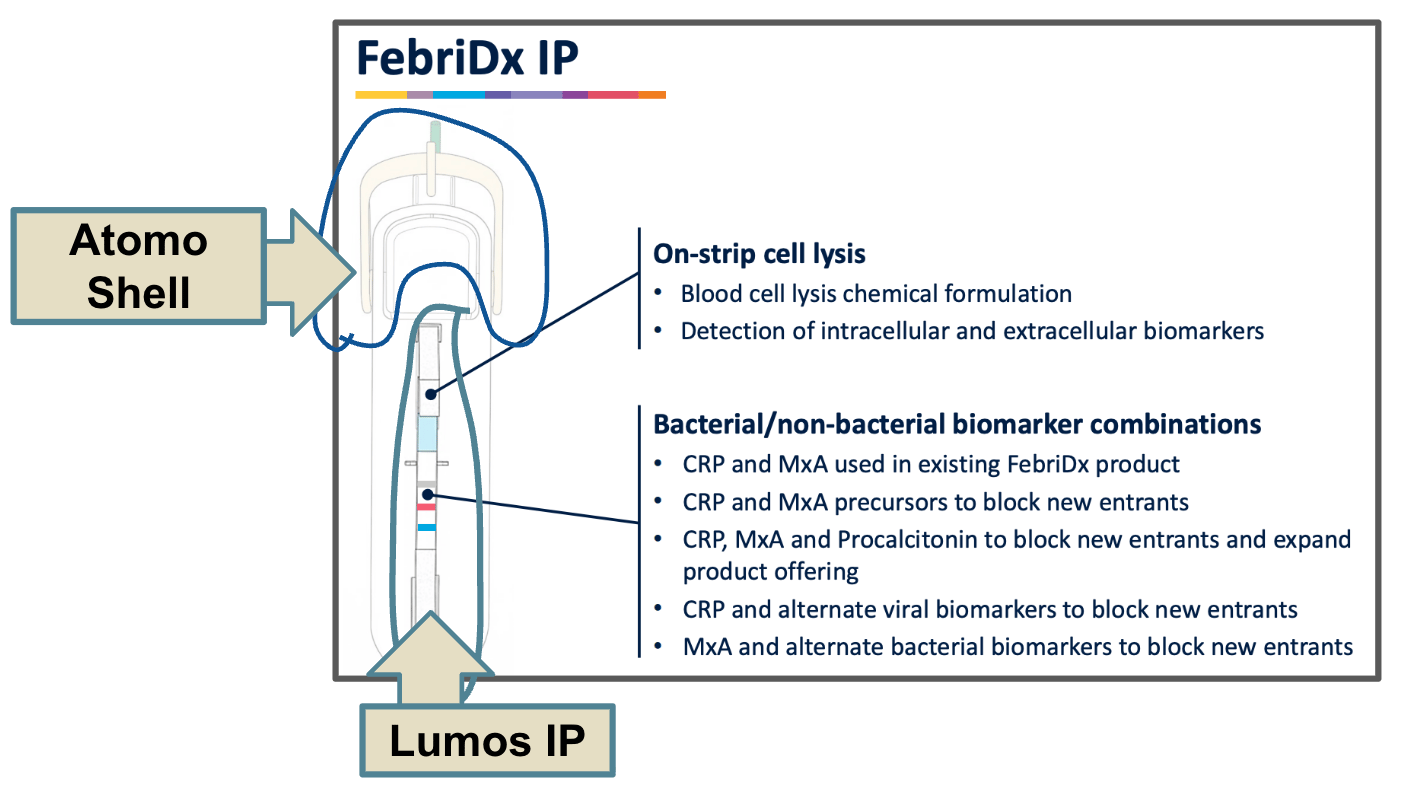
So Lumos is actually a customer of Atomo (and a big one at that).
If Lumos were to sell many of these devices, it stands to reason that Atomo would benefit as well.
In its response to the market, AT1 does not know the results of the FDA CLIA decision.

(Source, AT1 Response to ASX Price Query)
But the stock still traded up that day.
It’s an interesting way for insiders to play the Lumos Diagnostics result without trading in the company directly.
(or the volume could just be speculators coming in, who knows)
Interestingly, Atomo moved into a trading halt this morning.

(Source, AT1 Announcement)
Even if they don’t know anything, people trading their stock might.
So it's the safe play.
Over the last two weeks, the biotech industry has been one from two when it comes to binary results.
(Immutep bad, Amplia good)
A successful CLIA waiver application could make it two from three.
Trading halt lifts tomorrow.
Good luck!
The Armchair Analyst.





