Good morning,
Welcome to today’s edition of The Armchair Analyst, a 5-minute daily update on the ASX life-sciences sector.
Great science doesn't always make a great business.
The difference? The value proposition.
But if you’re not a doctor… how do you actually evaluate a medical product?
Companies tell us the answer, of course.
But we don’t actually know, for example…
What surgeons need at 7 am in an operating theatre.
What makes a procurement manager say yes.
What the rep said in the hallway that closed the deal.
So we validate it.
Is there a real problem? Is the solution actually good? What would it take for someone to buy this product?
These are the questions that matter, and for most of us, they're hard to answer just from reading through company announcements.
So for this next company in my Biotech 165 Challenge, I did something different.
I enlisted a surgeon to answer one question: What actually makes you buy a product?
Today, we are using Orthocell (ASX: OCC | $200M MC) as our guinea pig.
Because, well, my surgeon friend likes their nerve repair product, and that’s the one he chose.
(He also owns some shares in the stock)
Big thank you to Surgeon Dan for lending his expertise to this one.
Over to you, Dan.
Hi Surgeon Dan here,
I am a doctor who has been working in urology for the past 7 years.
I’m also a big stock investor.
Urology is a surgical specialty focused on diagnosing and treating diseases of the urinary tract (kidneys, bladder, ureters, urethra) and the male reproductive system.
Last week, I was asked, "What actually makes me buy a product?"
I’ve bought and used many medical products over the years.
Some good, some not-so-good, and some great ones that stand the test of time.
But what makes for a good product?
Here’s what I’ve learned.
Getting a product sold in Australia.
First, the product needs to be approved.
Approval is granted by the TGA after the company provides sufficient evidence to demonstrate that the product is safe and effective.
(Then it gets listed on the Australian Register of Therapeutic Goods)
This is a license to sell, but doesn’t guarantee sales.
To sell an approved product, each company enlists product sales representatives (known in the business as reps) to engage with hospitals and clinicians to get their product off the shelf and used in patients.
Think of the reps as the troops and the clinicians as the gatekeepers.
The reps come through, give their pitch (on occasions multiple times) and will look to schedule a product demonstration.
What does a product demonstration look like?
Surgeons trial products during live procedures, guided by the rep standing right there in the room.
Try before you buy.
The reps receive direct feedback from surgeons during the procedure. What works and what could be improved.
I've had my fair share of demonstrations.
The good, the bad and the ugly!
What makes a surgeon buy and use a product in Australia?
I’ve put together this framework for how I personally evaluate the products I buy and use, from those that I don’t.

1. Does the product improve patient outcomes?
New products must demonstrate that they address a significant area of target patient outcomes and markedly improve them, enabling a shift in practice.
This is demonstrated through:
Clinical research papers and peer-reviewed journals (yes, we do read them)
Actual evidence from our patients and what we see when they leave the surgery
If our patients improve, we’ll continue to buy the product.
2. Is the product fast and easy to use?
In surgery, time matters.
It is well recorded that any operation beyond 5 hours holds a significantly higher risk of general wound infections, deep vein thrombosis (DVTs) and pneumonia.
Every 30 minutes of sleep increases patients' risk of complications by about 10 to 15%.
Even the best product needs to be deployed quickly and easily. The longer a patient is under anaesthesia, the more difficult the recovery.
I avoid products that are difficult to use or time-consuming to set up and prepare.
3. What is the cost of the product?
This depends on whether the product is or isn’t covered by Medicare or a Health Fund.
IF COVERED: Generally, the surgeons will be cost-agnostic; if the product helps, then we will buy it.
IF NOT COVERED: We have to decide if the cost of the product that will be passed on to the customer will affect their decision to take up the procedure. If it's too cost-prohibitive and there's no coverage, we may offer it to clients but not include it as part of our general service.
4. Is the product easily accessible and available?
It’s all well and good to know that the product exists…
But if it’s not on the shelf when I need it, then I’ll likely use something else.
5. Surgeon Preference
Numbers 1 to 4 are the baseline for any product to be used by a surgeon.
But once the product ticks all those boxes, it then comes down to the surgeon’s preference.
Getting surgeons to switch products can be hard.
We get used to doing procedures in our own way, with our own product set.
So new products that come to market looking to displace existing products, can’t just be equivalent.
They need to be markedly better for a surgeon to make the switch.
Alternatively, if there is a true unmet need, then adopting those products is a much easier decision.
That is what I saw when two Orthocell (ASX: OCC) reps came into my practice three weeks ago to demonstrate Remplir’s ability to restore erectile nerve function for men who have had prostate cancer.
Because this is a true unmet need.
This next part is not financial advice. I do own some shares in Orthocell.
What’s the story?
Orthocell (ASX: OCC | $200M MC | $50M Cash) is an ASX-listed tissue-engineering company focused on regenerative medicine; its flagship product is Remplir.
Remplir is a collagen matrix used to facilitate peripheral nerve healing during surgery.

Derived from porcine peritoneum (the scientific name for the abdominal lining of a pig), it undergoes a proprietary process to render it inert to the body's immune response and has been proven to contain no residual genetic material.
This is an important safety step.
I first became interested in the product (and the company) when two reps from Orthocell came in to demonstrate Remplir.
There is a specific surgery for men with prostate cancer called a Nerve-sparing Robotic Assisted Radical Prostatectomy (NS-RARP).
It is a high-precision surgery used to treat localised prostate cancer by removing the prostate gland while meticulously preserving the delicate nerves and blood vessels responsible for erectile function and urinary control.
During the surgery, the neurovascular bundle (NVB), which lies on both sides of the prostate, is dissected away from the prostate and preserved to maintain erectile function and continence.
Orthocell is now using Remplir to help heal these nerves post-surgery.
Even with advances in robotic technology, the rates of impotence following nerve-sparing procedures can be as high as 80%.
I see this Remplir product as a ‘game changer’ in this space.
I’ve already adopted it in my practice.
Remplir has secured TGA clearance for use in peripheral nerve injuries in March 2022 and FDA clearance in March last year.
It has been marketed as a peripheral nerve repair product for Orthopaedic and Plastic Surgeons; however, it has some competition in those areas.
In urology (where I work), this is a genuine unmet need.
The best alternatives are nerve regeneration products derived from human donor cells.
These have demonstrated a 2.5-month reduction in recovery time, but are not widely available.
(Remember point 4 Access and Availability)
Remplir is derived from porcine peritoneum (pig cells). It is a raw material that is abundant.
Manufacturing is therefore scalable, and importantly for me, the product will be available when I need it.
Results from Remplir’s erectile potency and continence study are due in 2026, based on an initial rollout into the urological sector in America and Australia.
This is ‘real world data’ that I like to see (not cherry-picked patients for a clinical trial with exclusion criteria that may not match real life).
Here was the quote from the latest quarterly report:

(Source, Orthocell 4C Report)
I was one of those 100 studies conducted so far.
I’m hoping they will use the simple but standardised Sexual Health Inventory for Men (SHIM) score to demonstrate effectiveness pre- and post-Remplir.
It will underpin future studies that could lead to it becoming the standard of care in NS-RARPs.
(Which would be a big success)
My experience using the Remplir product
I deployed the product a few weeks ago, and it was easy.
Open the package, pick it up, place it, done.

The whole process took about 1 minute. The number of units used per case varies, but we used 4 for this case (2 on each side of the NVB).
Multiple sizes are available at different price points to suit the operation's needs.
So, how does Remplir’s product stack up on my framework?
1. Does the product improve patient outcomes?
In December 2024, Orthocell published the results of a pivotal study that showed that its product was safe and effective for the use of surgical repair of peripheral nerve:

While this study showed the return of voluntary movement in several indicators, Orthocell has yet to publish data on NS-RARP.
(The area I’m most interested in)
There have been ~100 surgeries conducted so far, and Orthocell will release data from these surgeries once it is compiled.
Positive results here will go a long way toward proving that this product improves patient outcomes, as there is no other product in the NS-RARP space that targets intra-operative potency without downside risk.
2. Is the product fast and easy to use?
The product was easy to use.
Drop in and done, no teething issues. Certainly hit the speed brief.
3. What is the cost of the product?
Remplir is fully reimbursed in Australia.
So I am cost-agnostic.
In America, the product is priced better than any competitor's (human cell stuff), which would help adoption in that region.
4. Is the product easily accessible and available?
Remplir has already been stocked in multiple hospitals nationwide as a result of its rollout in Orthopaedic and Plastic Surgery.
The company has sorted production and even ramped up plans with automation in mind.
It is on the shelf, ready to use.
The company currently has the capacity to produce 100,000 units annually and locally in GMP facility in Western Australia, with a long 3-year shelf life.
(Here’s the photo)

No special storage requirements, lightweight packaging and easy to ship worldwide.
Tick, tick, tick.
My Final Thoughts
From personal experience, Remplir in the NS-RARP environment can significantly improve patient outcomes.
It takes minimal additional time and does not change how the operation is done.
This product created a bit of a buzz with my colleagues, with four other surgeons keen to trial it.
This type of organic growth is where I have seen products take off in the field.
According to the company, this is a US$1.6B – US$2B opportunity in a category with true unmet need.
As an adjunct, it has all the upsides, and there has never been a recorded adverse outcome with this product.
Let that sink in.
No adverse outcomes recorded.
As a doctor who loves ASX investing, I like to dive deep into the companies behind the products I use and the innovations that come through my practice every day.
Just like Orthocell.
If you have any questions about the product or the company, feel free to reply to this email, and I’ll make sure to answer to the best of my ability.
The Armchair Take on Orthocell
Firstly, thanks, Surgeon Dan.
That was a very helpful framework to understand companies selling medical products in the field.
Now, for my take on the company from an investor lens.
Orthocell is in an interesting position.
This is not Orthocell’s first product in the market; it also sells a dental regrowth product and a cartilage regrowth therapy.
But right now, a bet on Orthcoell is a bet on Remplir nerve repair.
18 months ago, the company’s stock went on a mighty run after securing FDA clearance to sell Remplir into the US Market.
This was a genuine re-rating event for the company, and it gained momentum as it entered new markets and generated initial sales.

Off the back of this re-rate, the company raised $30 million at $1.30 per share, and the stock has been in a downward trend since.
Why?
There is a lag between when companies move from pre-revenue, catalyst-driven stocks to revenue-generating sales companies.
Ramping up sales takes time, and investors who were ‘in it for the catalyst’ churn out of the stock.
I call this the baton pass.
The baton is passed from the retail punters who like to play the catalysts to institutional investors who back businesses based on longer-term value.
Institutional money can be slower to make decisions.
They may want to see several quarters of revenue growth before building a position.
So right now, the Orthocell “bet” is on its ability to turn Remplir into a big revenue generator.
But as I’ve learned from Surgeon Dan… products can take time to become staples in a surgeon's toolkit.
That said, Orthcell is well funded (with A$50 million in the bank) to deliver on the promise to move up the adoption curve and convince surgeons beyond the early market adopters:
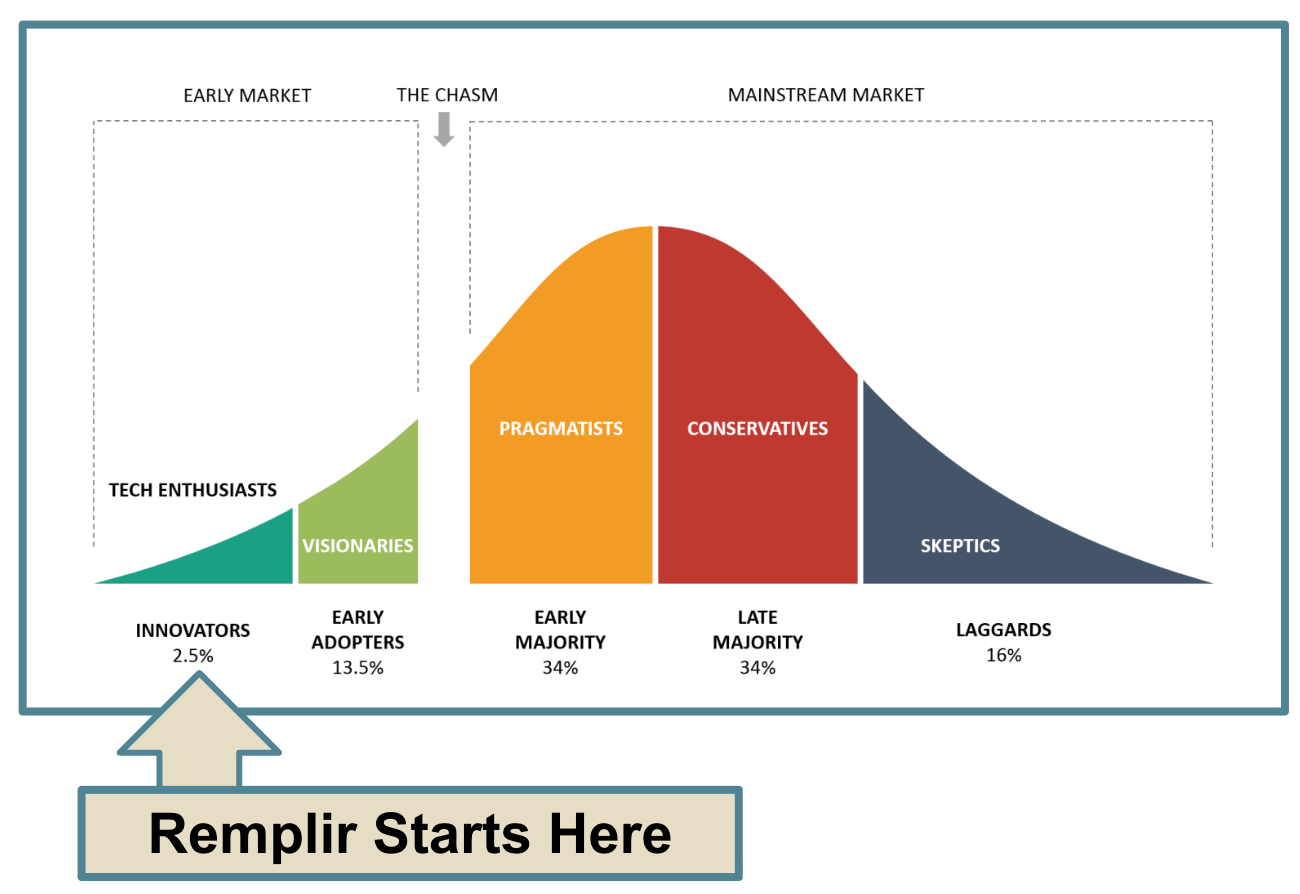
Over the last 12 months, Orthocell’s risk has shifted from regulatory (getting the product approved) to execution (getting the product sold).
In that scenario, it is a race to breakeven.
This creates an opportunity for traders to speculate on how quickly sales will ramp up.
Given the dip in the share price following the $30 million capital raise in August last year, the market is starting to price in longer timelines.
But keep an eye on those quarterly and half-yearly reports.
Any sign of revenue momentum should be rewarded by the market.
So, what’s the “bet” now?
Now, the bet is that Orthcell can start selling its product in the US market, with new indications like NS-RARP as the blue-sky upside for the stock.
The main risk is adoption.
But if the customer segment is anything like our friend Surgeon Dan, then product uptake may be quicker than the market expects.
A big thank you to Orthocell Managing Director Paul Anderson for speaking with Dan and me last week and sharing the company story.
See you all tomorrow,
The Armchair Analyst.
But first…
The Pulse Check
Radiopharm Theranostics (ASX: RAD) achieves concordance between its cancer imaging product, RAD 101, and MRI for brain metastases in 18 of 20 patients so far in the Phase 2b trial. (RAD)
🪑 First interim results published in December last year showed 11/12 patients achieving concordance with the cancer imaging product. 10 more patients to go to get the full trial results. On track to meet the primary endpoint.
BlinkLab (ASX: BB1) commences pivotal FDA 510(k) validation study for its autism diagnostic aid product. (BB1)
🪑 Pilot study showing 83.7% sensitivity and 84.7% specificity, this is the benchmark. FDA submission expected at the end of 2026.
Emyria (ASX: EMD) confirms its first Victorian psychedelic therapy clinic is on track to launch in Q2 CY2026. 31 therapists have been contracted and trained, with 4 psychiatrists also trained up. (EMD)
🪑 One of the bottlenecks for EMD is to scale up a skilled workforce of therapists and psychiatrists to deliver the psychedelic-assisted mental health treatments. Milestone ticked.
Cyclopharm (ASX: CYC) secures a multi-site agreement with Northwestern Medicine, including one immediate installation of its lung imaging device Technegas, and the potential to expand into six additional sites. (CYC)
🪑 Two days, two deals. Nice momentum.
Indian generics companies, including Sun Pharma and Dr Reddy's, launch cheaper versions of the weight-loss drug semaglutide as Novo Nordisk's patents expire in the region. (Fierce Pharma)
Cash Injection
Former Google subsidiary Verily raises US$300M to accelerate AI and personalised healthcare software. (Fierce Biotech)
Private, venture-backed Earendil Labs raised US$787M to scale its AI-driven biologics platform. (PR Newswire)
🪑 The chequebook is open right now for healthcare companies in AI.





