Good morning,
Welcome to today's edition of The Armchair Analyst, a 5-minute daily update on the ASX life-sciences sector.
A sensational 9 years.
A rocky 12 months.
… and now things are starting to recover.
The next company on my Biotech 165 Challenge is the second-largest biotech on the ASX.
Today, I’m covering Telix (ASX: TLX).
Telix has a radiotheranostic platform for the diagnosis of prostate cancer.
It is set to generate US$950-$970M in sales this financial year.
In the last week, Telix has signed a US$2.1 billion deal with the NASDAQ-listed Regeneron.
This validated radiopharma as no longer just a platform curiosity but as the next big bet for pharma in oncology.

(Source, Fierce Biotech)
The promise of radiopharma is targeted cancer diagnostics and a potential treatment platform.
There are three major selling products in the space.
Novartis (US$243 Billion), Lantheus (US$5.4 Billion) and Telix (A$5 Billion)
Because Telix is one of our flagship biotech stories on ASX, I’ve once again enlisted the help of Surgeon Dan to bring his clinical perspective on Telix.
He has first-hand experience with Telix’s product and understands its limitations and opportunities.
Big thank you to Surgeon Dan for lending his expertise once again.
But first…
The Pulse Check
Memphasys (ASX: MEM) reports its first $100,000 in revenue from sales of its sperm separation system, following its CE Mark approval earlier in the year. (MEM)
🪑 Good early traction, however, cash balance sitting at under $500,000 - I expect some new financing news soon.
Orthocell (ASX: OCC) has the first successful surgery with the Remplir nerve repair product within the US Veterans Affairs hospital network after announcing a deal yesterday. (OCC)
Nexalis Therapeutics (ASX: NX1) advances Phase 1 trial for a cannabis inhalant for panic disorder. Cohort 1 dosing completed (8 patients), and Cohort 2 commencing after approval from the safety review committee. (NX1)
🪑 Milestone ticked.
Immuron (ASX: IMC) reports Q3 sales of AUD$1.5M for its Travelan travel diarrhea product. (IMC)
🪑 On track to match last year’s sales revenue.
Proteomics International (ASX: PIQ) announces a strategic restructure, 25% of positions made redundant ($1M cost savings), and the retirement of Chairman Neville Gardiner. (PIQ)
The FDA has requested additional safety data from Eli Lilly's newly approved weight-loss pill, Foundayo. (Reuters)
🪑 There is a great LinkedIn post that explains the difficulty of delivering oral peptides in pill form.
This helps put into context my article from yesterday about Tetratherix’s (ASX: TTX) nasal spray technology for peptide drug delivery.
(I own 16,250 escrowed TTX shares)

(Source, LinkedIn)
Cash Injection
NASDAQ-listed Revolution Medicines has raised $2 billion through a combination of stock and convertible debt (Endpoints)
🪑 This markets the largest public financing for the biotech industry since the COVID-19 pandemic.
Here is where the US firmly sits on my capital waterfall chart:
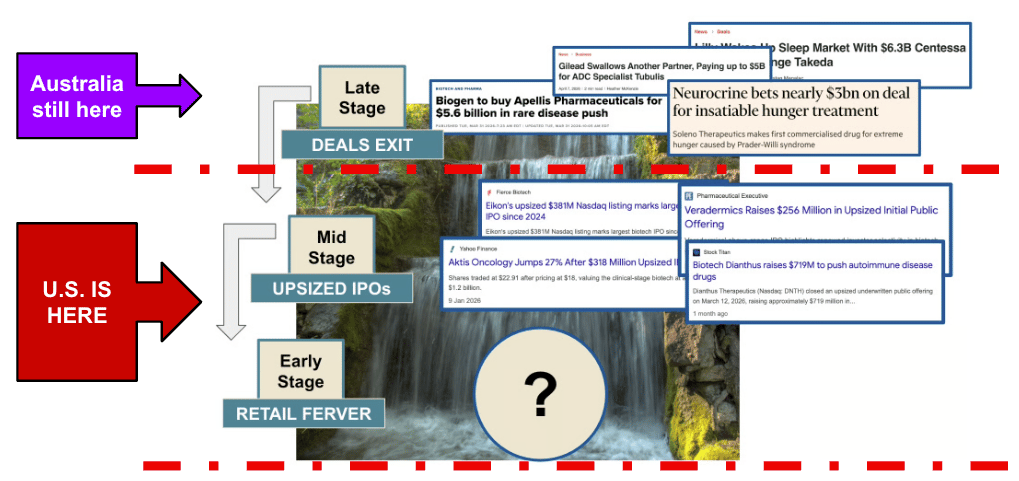
Australia still needs a few more late-stage deals to kick things off for the sector here.
Event Spotlight
If you’re in Melbourne next Thursday afternoon at 3:45 pm, come say hi!
I’ll be introducing the companies at the Monsoon twilight briefing; there are some good ones presenting:
Neuren (ASX: NEU)
Alterity (ASX: ATH)
Argenica (ASX: AGN)
Confirm your interest 👉 RSVP

Under the Microscope
Hi Armchair Army. It’s Surgeon Dan here.
I am a doctor who has been working in Urology for the past 7 years.
(and a big stock investor)
Last time on the Armchair Analyst, I introduced my framework for how to evaluate the medical products from a “Surgeon’s Perspective”.
(using Orthocell as an example)
Today, I will apply that same framework to Telix Pharmaceuticals (ASX: TLX), a company whose product I interact with almost every single week.
What’s the story?
Telix is focused on the development and commercialisation of diagnostic and therapeutic radiopharmaceuticals (theranostics).
Their ethos is straightforward…
If you can see it, you can treat it.
The field that Telix is in is also known as “nuclear medicine”.
Nuclear medicine is a specialty that uses small amounts of radioactive substances to diagnose and treat diseases.
Essentially, a small amount of a radioactive tracer, like Gallium-68 or Fluorine-18, is injected into the patient.
The tracer travels through the body and is taken up by highly active cells, such as cancer cells.
A PET scanner then detects this radioactive material, creating detailed images that show exactly where the cancer is.
Light up the cancer like a Christmas tree.
Telix sells the tracer and is currently generating revenue.
In its last quarterly report, Telix guided FY2026 revenue between US$950M and US$970M.

(Source, TLX Q1 Business Update)
The flagship products are Illuccix and Gozellix, both for the diagnosis of prostate cancer.
But they also have a long pipeline covering multiple cancers over the next decade:
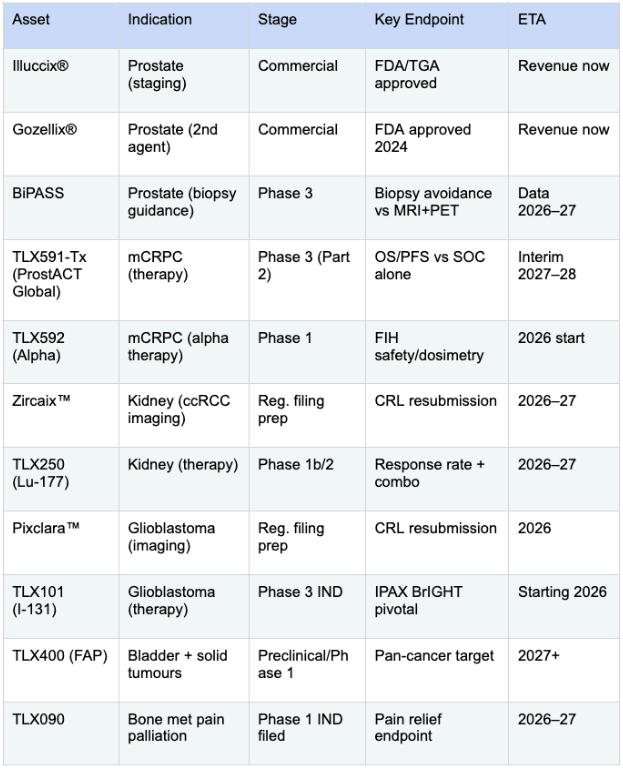
So, that’s a quick overview of the company.
How Nuclear Medicine Changed Patient Management… Fast
For almost ten years, I have been part of a weekly meeting of medical professionals from different disciplines to reach consensus on the diagnosis and management for high-risk cancer patients.
These meetings involve oncologists, radiologists, nuclear medicine physicians, allied health professionals, and, of course, surgeons.
Over the last ten years, I have seen how nuclear medicine has changed patient management and improved cancer diagnostics.
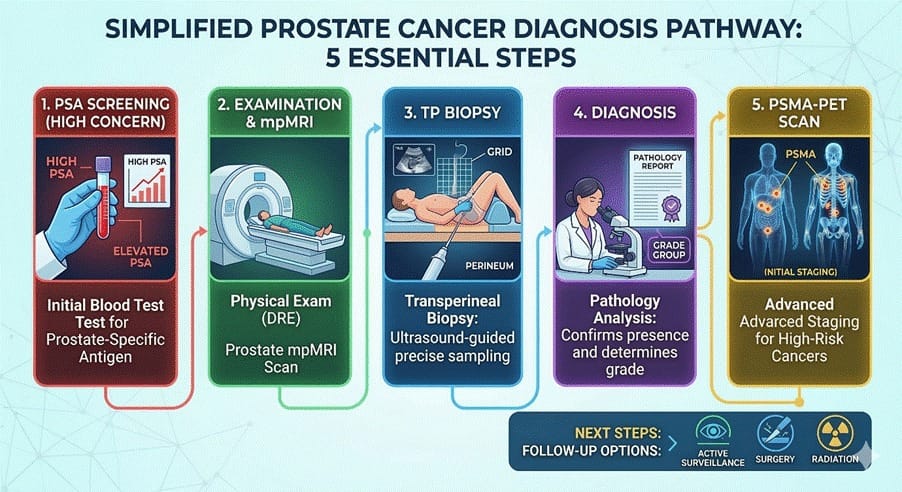
Using prostate cancer as an example (my specialty)...
Before nuclear medicine, we would conduct a full-body CT scan and a bone scan to evaluate whether a patient had cancer.
This led to a high false-positive rate and frequently over-staged patients.
An over-staged patient is someone whose cancer has been assessed as more advanced or widespread than it actually is.
This can lead to unnecessarily aggressive treatment.
So the patients were not getting the right care.
PSMA-PET was initially slowly rolled out in the late 2010s, but we could see how impressive it was and it changed the game.
The proPSMA study published in 2020 found that PET scans had 92% accuracy compared with 65% for CT/bone scan in the staging phase.
These are the kinds of numbers that change clinical guidelines and practice worldwide.
Simply put, the ability to clearly identify where the cancer is allows the clinician to treat it much better.
This is the basis of the Telix philosophy, and it underscores everything they do.
What I’m most looking forward to in Telix’s pipeline
Telix has a large product pipeline, but there are a handful of programs I think are the most interesting.
1. BiPASS: Changing when a surgeon scans
A Phase 3 clinical trial to expand the use of its product before a biopsy.
The goal is to eventually eliminate the need for tissue sampling in certain patients.
We (surgeons) request PSMA-PET staging scans for virtually all intermediate and high-risk patients.
That’s the current standard.
But what if we could scan patients earlier?
Before the biopsy, alongside the MRI, and potentially remove the need for a biopsy altogether?
This would improve patient comfort, maintain diagnostic accuracy and reduce the need for an additional procedure with potential risks…
That’s the value proposition of BiPASS.
(Get it… bypass the biopsy… BiPASS)
Now, a reality check.
It would take a lot of evidence to shift a surgeon’s concern about removing a prostate without tissue confirmation first.
The implications of operating on a patient who doesn’t actually have cancer are serious.
So we tend to remain on the side of caution.
If the evidence stacks up, however, Telix could capture a much larger market.
All potential prostate cancer patients, not just those already confirmed as intermediate or high-risk.
2. Zircaix: A Paradigm Shift for Kidney Cancer
Zircaix is a Zirconium-based PET imaging agent that targets a protein expressed on over 95% of the most common type of kidney cancer.
In a study of almost 300 patients, Zircaix demonstrated a positive predictive value (PPV) of 92.9%.
This wasn't just published in any peer-reviewed journal; it was in The Lancet Oncology (a big deal in medical circles).
Also, 48.6% of patients had a change of management based on the scan results.
To put this in context, the current standard is a multi-phase CT scan, which has a PPV of approximately 59 – 67%.
(And it is not as accurate for small tumours under 4 cm).
This is once again a major topic of discussion in our weekly multidisciplinary meetings among medical professionals. How do we properly diagnose small renal masses?
Right now, it is based on features within the CT scan, but there are no specific uptake markers to confirm its accuracy.
This leads to either a delay in diagnosis or over-treatment.
The vast majority of kidney cancers are asymptomatic and incidentally identified due to a scan being done for another reason.
Every year, 400,000 kidney cancers are identified.
Approximately 20% of ALL abdominal imaging identifies kidney lesions (which leads to more scans).
Those scans?
The only answer we have is a CT multi-phase scan.
Sound familiar…
This reminds me exactly of the paradigm shift we saw in prostate cancer.
We went from CT plus bone scan (65% accuracy) to PSMA-PET (92% accuracy) and it changed clinical guidelines in prostate cancer worldwide.
Zircaix has the potential to do the same thing for kidney cancer, from 67% to almost 93% PPV.
This is clinically significant and, in my view, will change how renal cancers are imaged going forward.
FDA resubmission response is expected in 2026–27 following an initial Complete Response Letter to address supply concerns – not about the clinical data.
The ZIRCON Phase 3 results were never questioned; it was how Telix would actually supply the isotope.
(We’ll revisit this in challenges below).
3. ProstACT Global: Curing Prostate Cancer.
The idea is not just to be a diagnosis but also a therapy.
This is the blue sky.
TLX591 is a first-in-class radio-antibody designed to treat metastatic castration-resistant prostate cancer (mCRPC)
(the most advanced and difficult-to-treat form of prostate cancer)
The current market leader is Novartis’ Pluvicto, which reached blockbuster status in 2024 with US$1.4B in sales.
TLX591 differs because it is designed to remain within target cells for weeks rather than days.
Larger gaps between treatments and better tolerance for patients.
ProstACT Global is currently in Phase 3.
Part 1 has been completed. Part 2 is a randomised global multicentre trial of approximately 490 patients, with interim data expected in 2027–28.
The treatment would likely be priced similarly to the Novartis product at approximately US$42,000 per course.
Safety has been confirmed. Now it’s about the final efficacy result in Part 2.
My Surgeon’s Scorecard
Last month, I introduced a 5-point framework for evaluating medical products.

So, let’s see how Telix stacks up.
1. Does the product improve patient outcomes?
PSMA-PET scans have fundamentally changed how we, as surgeons, manage patients.
Particularly in prostate cancer.
So, tick.
2. Is the product fast and easy to use?
Injectable kits are easy to administer. Same-day scans with quick turnaround.
Each product in Telix follows the same proven system.
Another big tick from me.
What is the product's cost?
Both Telix’s flagship products, Illuccix and Gozellix, have secured reimbursement in Australia and the US.
This means that as a surgeon, I am cost-agnostic.
Tick again.
Is the product easily accessible and available?
Alright, this is where things get a bit challenging.
The single biggest factor limiting Telix’s wider adoption is the limited half-life of the underlying isotope used in the PET imaging.
Gallium-68 has a 1-hour, ultra-short half-life.
(half life means the time that it takes to degrade and become unusable)
So, to get the product on time and into the hospital, an expensive distribution infrastructure is required.
Cyclotrons to produce the radioactive isotopes and a network to distribute the final tracer doses to hospitals.
Also, it limits access and the number of scans that can be done per day, per centre.
Right now, access to PET scans is limited (and needs to be booked well in advance).
This makes effective triage necessary to prioritise only the highest-risk patients for scanning.
While the current situation is manageable, it is far from ideal.
One of my jobs as a junior doctor was to call all the nuclear medicine centres across the state to see how quickly they could squeeze in another patient.
In one way, it shows the limitations.
On the other hand, it demonstrates the overwhelming demand.
(PET scan availability is the surgeon's equivalent of a Taylor Swift concert ticket).
Telix has been proactive here.
They acquired RLS (RadioLigand Solutions) in January 2025 and are deploying their QIS cyclotron technology to achieve vertical integration and control the supply chain.
Surgeon preference?
PSMA-PET is essentially a worldwide standard, accepted in most, if not all, international guidelines.
It goes beyond ticking a box; this is essential to provide the best care for the patient.
So, four out of five on my surgeon scorecard for Telix’s product.
Nuclear medicine competition is heating up
I would be remiss if I wrote an entire article about Telix without mentioning another ASX-listed company, Clarity Pharmaceuticals (ASX: CU6).
As the current leader in prostate cancer staging, Telix is well-positioned and well-funded.
But Clarity Pharmaseuticals is hot on their heels.
The differences between the two products come down to the isotope.
While Telix uses Gallium (with a half-life of 1 hour), Clarity uses Copper (with a half-life of 12.7 hours).
This presents a significant solution to some of the logistical challenges Telix has encountered (and I’ve experienced as a surgeon using the product).
Also, in a head-to-head trial between Clarity’s and Telix’s products, Clarity demonstrated a greater ability to identify legions (more sensitive - identifying real cancer earlier and at smaller levels).
In the 50-patient trial, Clariy identified 63 legions, while Telix identified 24.
But right now, Telix is revenue-generating and can outspend Clarity. Addressing the supply chain weakness with infrastructure and acquisition deals.
But the fundamental physics of Gallium-68’s half-life cannot be changed.
It’s one to watch.
The Armchair Analyst Take
First, thanks again, Surgeon Dan!
With a $5B market cap and as the second-largest biotech company on the ASX, Telix is a beacon for the sector.
The revenues are hard to ignore.
US$950M - US$970M in FY2026 is a massive amount of funding Telix can allocate to its earlier-stage programs.
It's a deal with Regeneron completely validated the radiopharmaceutical industry and its strategy.
I think that the theory that Clarity is the Telix killer is overplayed - while they are in the same space, Telix has a first-mover advantage.
The catalysts to look out for include:
Label extension for the BiPASS trial
Resubmission of the Zircaix for kidney cancer (addressing some of the supply challenges that the FDA were concerned with)
Theranostics product for ProstACT Global
The bet for Telix is that its nuclear medicine platform is the next frontier of cancer therapy and diagnostics, and the partnership with Regeneron will create multiple blockbuster drugs.
It just has to overcome the logistical challenges and competition hot on its heels.
See you all next week,
The Armchair Analyst.





