Good morning,
Welcome to today's edition of The Armchair Analyst, a 5-minute daily update on the ASX life-sciences sector.
I have something special lined up for you today.
A guest post from Dr Berra Yazar-Kolinski, the founder and CEO of Yazar Lab and the former chief scientific officer at Lykos Therapeutics.
(The first company ever to go for FDA approvals for psychadelic-assisted therapies)
She has 17 years of experience running trials for psychedelic therapies… and effectively wrote the protocol for Lykos that Emyria’s treatment is based.
The topic we’re covering today is reimbursement.
What is treatment worth?
Psychedelic stocks in the US had a nice bounce today:
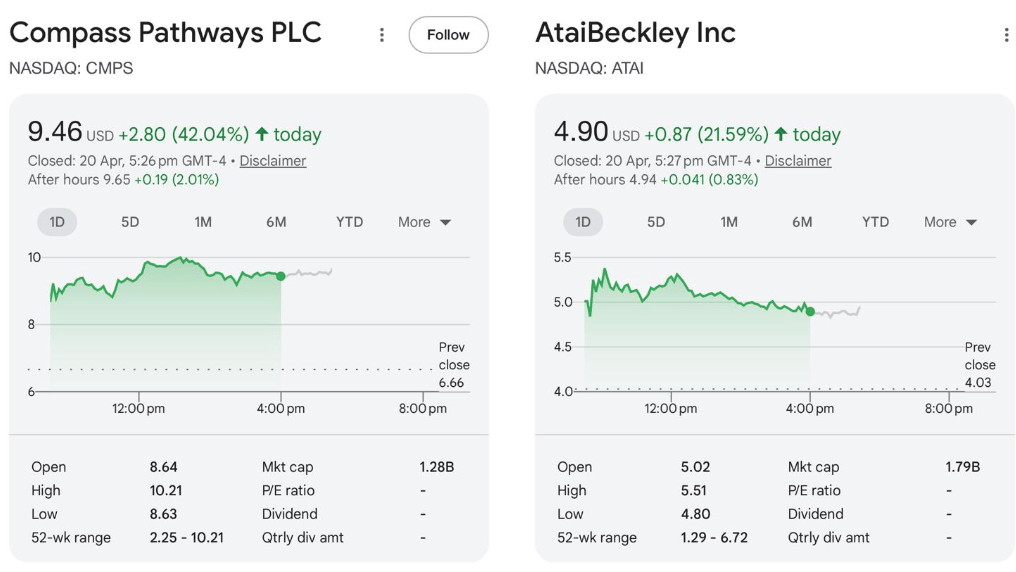
It is looking more and more likely that a psychedelic-assisted treatment will be approved in America within the next 12 months.
But FDA approval does not mean that it will be paid for.
A debate is underway in the US over what the reimbursement model should look like… and whether treatment should be treated as incremental improvement or transformational remission.
Dr Berra provides her thoughts on the topic…
While I tie it all back into my Second Armchair Analyst Pick for the year, Emyria (ASX: EMD).
Disclosure: I own 2 million shares in EMD.
But first…
The Pulse Check
Invion Limited (ASX: IVX) progresses its non-melanoma skin cancer trial to the third phase - Basal Cell Carcinoma patients - targeting the largest skin cancer market segment (80%). (IVX)
🪑 Nice, looking forward to these results.
Starpharma (ASX: SPL) completes FDA Type C meeting on clinical development plans for its HER2 radiotherapy candidate with its dendrimer payload delivery technology. Phase 1 study set to begin this quarter. (SPL)
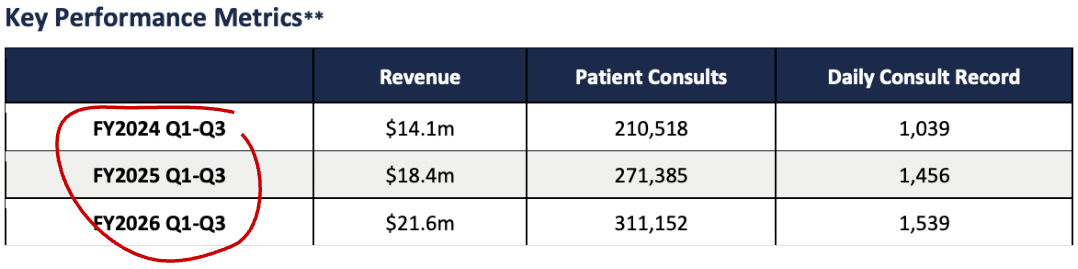
Vitura Health (ASX: VIT) reports that Doctors on Demand has surpassed 1M consultations. (VIT)
🪑 Interesting update, comparing the Q1-3 for each year.
I guess more information is better; it just looks weird to lop off that Q4 for each period.
AdAlta (ASX: 1AD) signs MoU with Oribiotech and Cell Therapies to deploy automated cell therapy manufacturing platform. (1AD)
🪑 I was trying to work out what each party's role was in the MoU.
Looks like Oribiotech owns the cell therapy technology. Cell Therapies has the GMP licence, and AdAlta will have a pipeline of cell therapy assets to apply the technology to.
The goal is higher throughput and shorter manufacturing timelines.
I don’t mind this deal - one of the biggest challenges with cell therapies is the manufacturing. If AdAlta has licensed a solution, it could be a point of difference for the company.
Australian Clinical Labs (ASX: ACL) is facing a class action in the Supreme Court of Victoria related to a 2022 cyberattack. (ACL)
🪑 Not good.
A major review of 17 studies finds anti-amyloid Alzheimer’s drugs like Leqembi and Kisunla offer little to no meaningful clinical benefit despite reducing brain plaques. (FiercePharma)
🪑 Actinogen (ASX: ACW) was up 17% yesterday on no news, and I suspect this circulating report had something to do with it.
Top-line results from the company's Phase 2b/3 Alzheimer’s trial are scheduled for November this year - and the current standard of care (anti-amyloid drugs) just took a big hit.
The Report Card
Bioxyne (ASX: BXN) reports record quarterly revenue of $21.3M (+137% YoY) and $1.2M in net operating cash inflow. (BXN)
🪑 Another very solid quarter for Bioxyne. Cash flow positive, on track for FY guidance of $65-75M
Compumedics (ASX: CMP) revises FY26 guidance, expecting revenue of $62M-$65M and EBITDA of $5.5M-$7M. (CMP)
🪑 This is a revenue downgrade; previous guidance was $70M in revenue and $9M in EBITDA.
M&A, Big Pharma Wants a Wife
Eli Lilly picks up another in vivo CAR-T company from Kelonia worth up to US$7B. (FierceBiotech)
UCB to buy Neurona in cell therapy deal worth up to US$1.15B for phase 1/2 cell therapy for epilepsy. (UCB)
Under the Microscope
Hi Dr Berra here,
I am the founder and CEO of Yazar Lab and the former chief scientific officer at Lykos Therapeutics.
I have run clinical trials for psychedelic therapies for 17 years and seen the full arc of drug development from preclinical to commercial planning.
The topic I am going to cover today is one I have been mulling over for a while.
This is because cost-effectiveness is the central tool for balancing risk/benefit with affordability, ensuring the greatest overall health gain from limited resources.
What are the true cost/benefits of psychedelic therapy, its potential and pricing for a PTSD recovery?
Compass Pathways is set to apply for FDA approval of psilocybin for treatment-resistant depression by the end of this calendar year.
Much has changed since we first attempted to obtain similar approval of MDMA therapy for Posttraumatic Stress Disorder (PTSD) through Lykos Therapeutics (now Resilient Pharmaceuticals).
Health Secretary Kennedy has been very in favour of psychedelic therapies, and the stars look to be aligning for a psychedelic therapy approved and on the market within the next 12 months.
Just this weekend, President Trump issued an executive order to speed up access to psychedelic drug treatments, including directing the FDA to issue Priority Review Vouchers to programs with breakthrough designation:
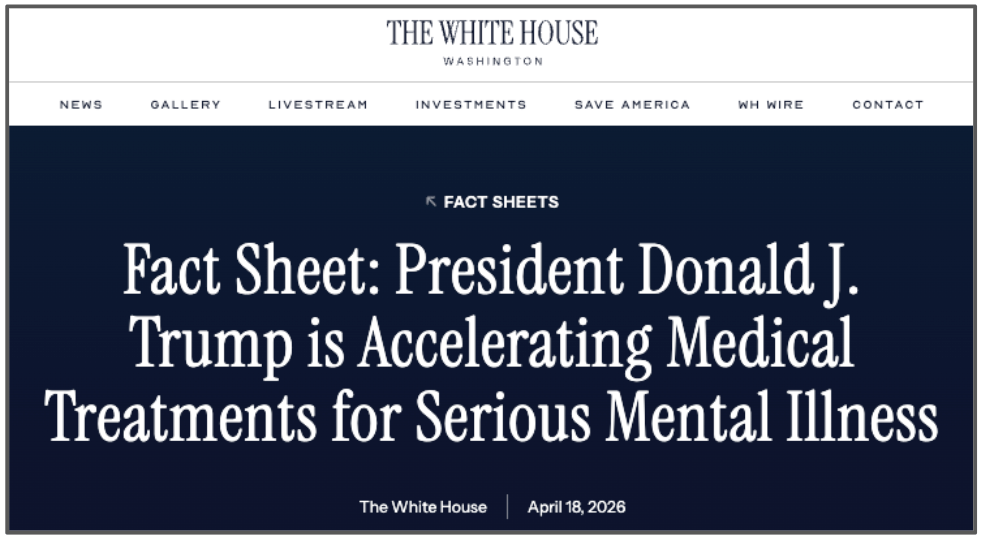
(Source: The White House)
While we are on the cusp of bringing psychedelic therapies to America, the healthcare landscape stands at a crossroads.
There is a great debate in the field on how to actually deliver psychedelic therapy.
Who will pay for it?
At what cost?
While health economics models provide a foundational baseline for value, they often fail to capture the full gravity of the disorder’s lethal potential and its broader societal impact.
Let’s take a closer look.
The Economic Case: Direct Savings and Untapped Benefits
Current modelling suggests that MDMA therapy is more cost-effective for chronic PTSD (moderate or higher severity) than standard available treatments.
But I see these figures as a conservative floor for their true value.
Here are the pricing considerations:
Modelled Cost-Effectiveness: Compared to Psychotherapy
A study published in PLOS ONE evaluated whether MDMA therapy is worth the cost compared to psychotherapy. (Source)
Over a five-year period, the treatment had an incremental cost-effectiveness ratio (ICER) of $83,845 per quality-adjusted life year (QALY).
In practical terms, that’s well below the commonly used $150,000 threshold insurers consider “worth it.”
So, MDMA therapy essentially works better, costs more upfront, but reduces future healthcare costs.
🪑 Armchair Analyst here - I just want to quickly explain QALY.
QALY stands for Quality-Adjusted Life Year.
It’s a metric used in healthcare and economics to measure the value of medical treatments by combining both the quantity and quality of life.
QALYs help governments and health systems decide which treatments give the most benefit for their cost. It’s a way to compare very different interventions on a common scale.
1 QALY = 1 year of life in perfect health
If your health isn’t perfect, that year is worth less than 1
Back to you, Dr Berra.
Comparative Cost-Effectiveness (in real-world settings)
Compared with standard treatment practices in specific healthcare settings, SSRIs have been modelled as cost-effective.
SSRIs are the drugs approved for the treatment of PTSD.
This is what MDMA therapy is effectively “competing against”.
SSRIs often yield lower QALY gains because of their primary role in symptom management rather than in remission.
A network meta-analysis identified EMDR (a type of therapy for trauma) as a highly cost-effective intervention, with a significantly higher lifetime QALY gain of 1.8 compared with no treatment.
Also, evaluated as a highly effective first-line intervention, TF-CBT (another type of therapy for trauma) has shown a mean QALY increment of approximately 0.12 in certain long-term models.
By these measures, MDMA therapy represents a more cost-effective treatment than either SSRIs or psychotherapies for PTSD.
Direct Savings
Over five years, MDMA therapy is projected to reduce expenditures on PTSD-related healthcare visits (-$2,511) and regular treatments (-$1,877) compared to therapy alone.
Notably, cost savings are not a requirement for a new treatment to be considered cost-effective.
But they play an important role in insurance companies' reimbursement decisions.
Societal Burden
Current models do not account for the broader societal burden of untreated PTSD, such as lost workplace productivity, family/caregiver strain, or the benefit of returning a patient to full societal participation.
The Mortality Risk: A Life-Saving Necessity
PTSD carries a risk of mortality that arguably exceeds many physical issues.
A critical oversight in standard economic discussions is the comparative lethality of mental health and physical health conditions.
Likewise, interventions for physical ailments are considered reimbursable at considerably higher rates, despite not posing a direct risk of mortality.
Premature Death: A PTSD diagnosis is associated with a 1.2-fold increase in premature mortality risk for US civilians and a 1.8-fold increase for the military population.
Suicidal Behaviour: Systematic reviews confirm that lifetime PTSD is a potent predictor of suicidal behaviour.
The Model Gap: Crucially, the "cost of mortality," the inherent economic value of a life lost, is not typically assigned a specific dollar value within standard cost-effectiveness models. This omission may lead to a significant undervaluation of treatments that effectively mitigate life-threatening psychiatric symptoms.
Final thoughts
With currently available treatments, PTSD is ‘managed’ and remains a lifelong diagnosis.
It is hard to find patients who say they “used to have PTSD.”
(Almost everyone on treatment still has it in some form or another)
Cost-effectiveness is the gatekeeper for adoption. But with psychedelic therapies, it likely understates the true value.
Psychedelic therapies may restore function in ways current frameworks fail to fully value, from productivity to family and societal impact.
By conventional metrics like the QALY, MDMA therapy already meets or exceeds accepted thresholds, delivering strong real-world and research outcomes and downstream savings.
But these models capture incremental improvement, not transformation - which MDMA-AT appears to demonstrate.
As approvals near, the question is no longer whether these therapies are cost-effective.
It’s whether our systems can recognise, and pay for, the full extent of their impact.
The Armchair Take
First, thanks so much, Dr Berra, for that take.
I completely agree that the transformational nature of psychedelic-assisted treatment should be reimbursed on the basis of remission, not incremental improvement.
Health insurance companies don’t bat an eyelid when it costs them $40,000 for a pacemaker, or $100,000 for a kidney transplant…
I see psychedelic treatment in this same conversation.
Particularly based on some of the results that Emyria (ASX: EMD) has shown.
(⅔ patients no longer have PTSD, 76% clinically meaningful reduction in PTSD)
Emyria has put together some modelling of the cost of its treatment ($30,000) vs the standard of care ($60,000).
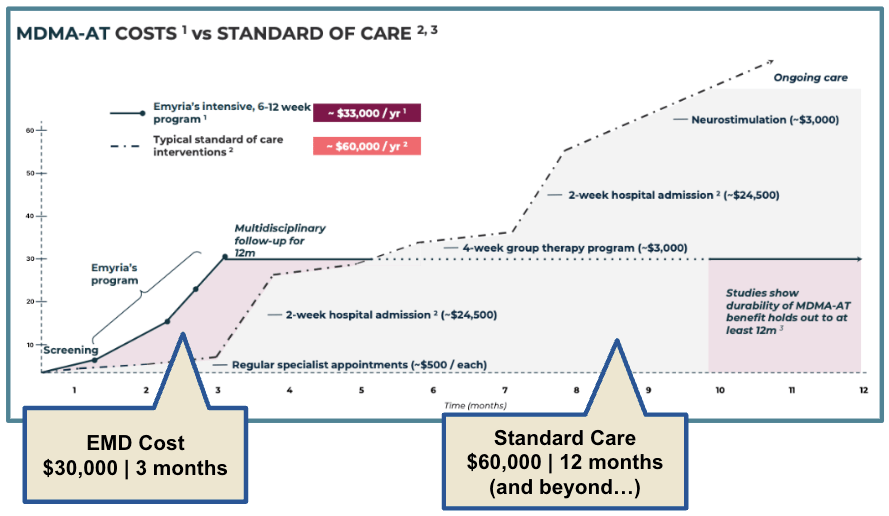
Where I think this analysis is flawed, however, is that it only benchmarks the standard of care for just one year.
Emyria has shown that the treatment is durable well beyond the 12-month mark.
This is called durable care.
If Emyria (and the broader mental health community) can demonstrate durable care.
1 year… 2 years…10 years down the track…
Then that $30,000 price tag that Emyria has placed on its services right now becomes incredibly cheap to a health insurance company.
But more data is needed…
Only a small number of patients have reached the 12-month mark post-Emyria’s treatment, and even fewer have reached the 2-year mark.
Over the next few years, Emyria will be able to build a comprehensive data package on the long-term effectiveness of its treatment.
Incredibly valuable data to health insurance companies and incredibly valuable data to US-based psychedelics companies with approvals on the horizon, having that “insurance coverage” conversation right now.
Using a value-based pricing model, I see this treatment is not worth just $30,000…
But potentially much higher if it actually is a cure for PTSD and other mental health issues.
Provided that the treatment WORKS in the long run.
Emyria is building a very defensible moat around its business and is the only organisation capable of delivering the service at scale in Australia.
This puts it in a strong negotiating position with health insurance companies as more long-term data emerges.
Right now, we are at the experimental phase.
Covering the cost for Emyria to make a margin and scale up the business.
But if Emyria can show lasting, durable results from its treatment, the sky is the limit for what it could become.
See you all tomorrow,
The Armchair Analyst.





