The next company in my Biotech 165 Challenge holds a special place in my heart.
It was the stock that got me into biotechs.
The first ever biotech I wrote about…
A true ‘trial by fire’.
Dimerix (ASX: DXB).
My old firm, Next Investors, decided to label Dimerix as its Biotech 2021 Pick of the Year on the Friday.
By Monday, we had our launch article ready to go.
The entire weekend I spent diving into rare kidney diseases, clinical trials, orphan drugs and big biotech exits.
Within an instant, I was hooked.
Much like many impulse purchases, you make the investment decision, and the true due diligence comes afterwards.
I go back and read through that initial article that I helped to write, and I think about how naive I was about biotech investing.
Dimerix is a bit of an enigma to me.
Just when you think you’ve figured it all out, you’re humbled.
Just when you’ve lost all hope, a rabbit emerges from the hat.
Up, then down, then up, then down.
Yesterday, Dimerix decided NOT to pursue accelerated approvals for its Phase 3 clinical trial on a rare kidney disease, FSGS.
The stock was smoked.
But the story is not over. Not even close.
Today, I put my first-ever biotech investment, Dimerix, under the microscope and try to explain why they made this decision and what it means for the company going forward.
They say that the best way to learn anything is to do it.
No textbook is ever going to teach you the emotional rollercoaster of investing.
Particularly early-stage biotech stocks.
Just when you think you’ve figured it all out, you’re humbled.
Just when you’ve lost all hope, a rabbit emerges from the hat.
There is always one stock that makes you think you’re a ‘genius investor’...
… and then shortly after, you’re back to your friends with your tail between your legs.
For me, that stock is Dimerix (ASX: DXB).
Dimerix is in the middle of a Phase 3 trial for a rare kidney disease called FSGS.
Yesterday was a bad day. The share price was down over 40%.
The reason?
The company decided against going after “Accelerated Approval” for its phase 3 clinical trial.
Here is the excerpt from yesterday’s announcement:

(Source, Dimerix Announcement 27th April 2026)
Since the start of the trial, “accelerated approvals” have been part of the investment thesis for many shareholders.
As with any share price movement, it's all about expectations and reality.
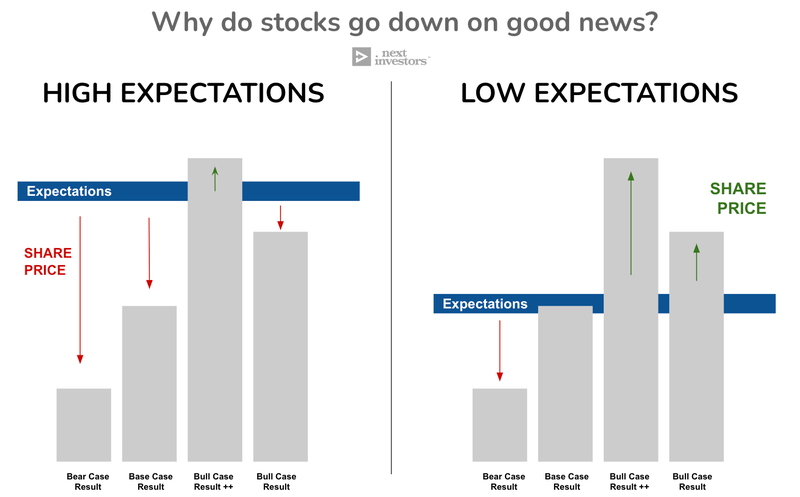
So, by deciding not to pursue accelerated approvals, the share price fell to reflect the additional 12-18-month delay in the drug approval decision.
Also, investors started looking at the cash balance and wondering if the $38.5 million that it had at the end of 2025 would be enough to carry it through the rest of the trial.
But why make this decision?
(Or as another friend put it to me, why the hell would they do that?)
The simple answer…
By NOT pursuing interim accelerated approval, Dimerix has made it easier to obtain full approval.
It’s all got to do with statistics.
(And I’ll explain more about it in a bit)
What's the story?
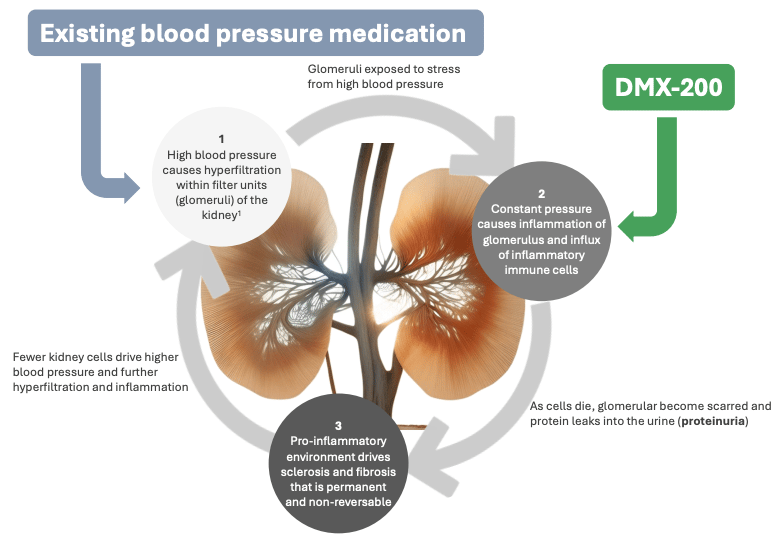
Dimerix is currently in a Phase 3 clinical trial for FSGS.
FSGS = Focal Segmental Glomerulosclerosis.
A rare kidney disease, where the tiny filters in your kidneys start scarring up.
Protein leaks into your urine. Kidney function falls off a cliff. The median time from diagnosis to end-stage kidney disease is 6 to 8 years.
There's no cure.
Until very recently, there wasn't even a single FDA-approved drug specifically for FSGS.
Enter DMX-200, Dimerix's lead drug candidate.
It is taken in combination with a blood pressure medication to restore kidney function.
The promise…
Reduce proteinuria, slow kidney decline, and give FSGS patients something that actually treats their disease rather than merely papering over symptoms.
As a rare disease, FSGS also has an orphan drug designation.
Which means extended periods of exclusivity, higher drug pricing and the potential for accelerated approvals.
The pivotal trial, ACTION3, is a 286-patient (later expanded to 333) randomised, double-blind, placebo-controlled Phase 3 study conducted across 219 sites in 21 countries.
Two-year treatment period.
The kind of trial that, if it lands, would represent the single most important data drop in FSGS history.
Right now, Dimerix has finished recruiting all of the patients in the trial, but still needs to complete the 2-year dosing regimen.
It secured a “go” for the interim futility analysis in early 2024 and has multiple licencing deals in place.
This is strong external validation of the trial and the product.
… and in biotech land, external validation is everything.
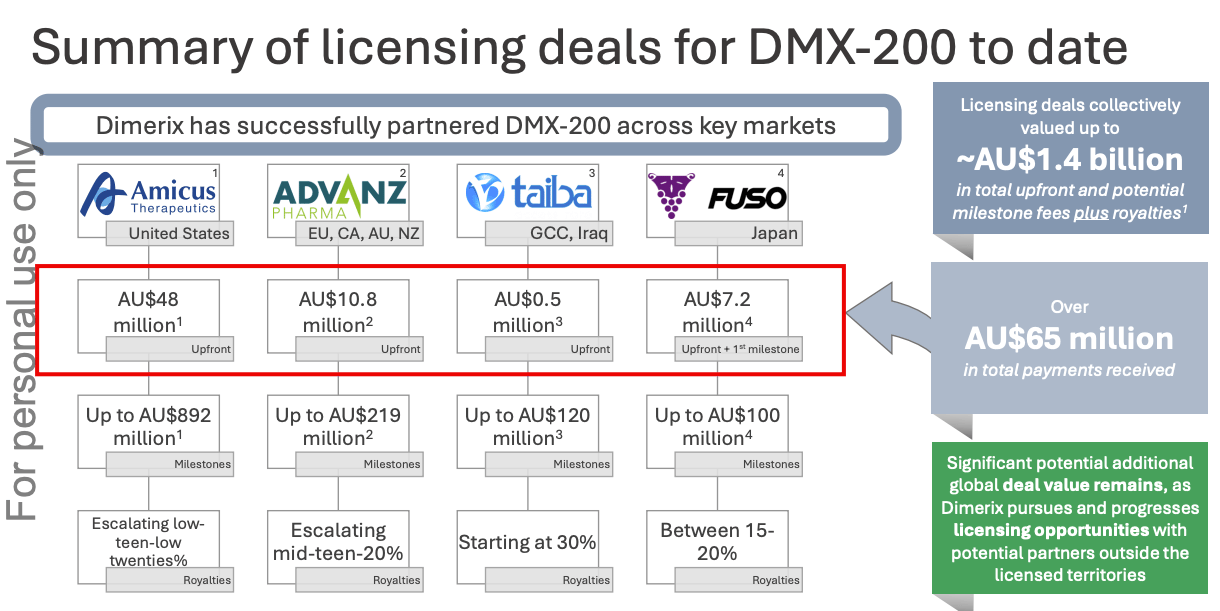
External Validation: Multiple licencing deals for Dimerix
The first deal (Advanz Pharma) came before the interim futility readout.
(It was a pretty epic deal, and completely wrongfooted the market…)
Dimerix was capped at ~$30M at the time, and they essentially received ⅓ of their market cap as an upfront payment (before any data was announced).

(Source: Dimerix announcement, October 2023)
Since that deal, it has secured three additional deals, locking up all major regions bar China.
The headline number is A$1.4 billion in upfront payments and milestones with additional royalties.
Where is the trial at?
Right now, Dimerix has completed recruitment for the trial.
333 patients randomised, dosing begun.
The last patient is expected to receive their last dose in March 2028.
Now, here is where it gets interesting.
Because FSGS has an orphan drug designation, Dimerix was eligible to apply for accelerated approvals.
Essentially, a faster path to market.
This was a big part of the investment thesis and part of the story going into the trial (particularly when I first covered the stock back in August 2021).
Accelerated approvals mean a faster path to market.
Even if the drug is approved on this basis, the company still needs to complete the trial in full.
(and if it fails the full analysis, then the registration could be pulled)
BEFORE Dimerix could go after accelerated approvals, there were four things that it needed certainty on:
FIRST, Enough patients completed dosing. Tick, this milestone was completed early last year.
SECOND, Consultation with the FDA. In March last year, the FDA provided a roadmap for both full approval and accelerated approval.
THIRD, Outcome of the PARASOL Collaboration Data. Tick, in October last year, the outcome of the PARASOL collaboration gave DXB a definitive path to accelerated approvals.
FOURTH, Powering Analysis. Tick, completed yesterday, which showed that the trial was sufficiently powered.
So all the pieces were in place for Dimerix to pursue accelerated approvals.
… yet it chose not to.
Before I dive into that decision, there is something we need to cover off.
As the Phase 3 clinical trial progressed, there were changes to the FSGS landscape, a working group called PARASOL was formed to solve a big problem…
Traditional endpoints for FSGS don’t work.
The changing endpoints for the FSGS trial
An endpoint is a specific measurement that researchers track in a study to determine whether a treatment or intervention has had any effect.
Until recently, the only proteinuria-based endpoint the FDA would accept for FSGS approval was complete remission (proteinuria <0.3 g/day).
Proteinuria = protein in the urine.
That's an incredibly high bar. Most patients in most trials don't get there, even on a drug that genuinely works.
The alternative was eGFR.
eGFR = Measuring kidney function decline directly.
But eGFR moves too slowly in FSGS to be practical. You'd need huge trials running for many years to see a difference.
In a rare disease where you can't even find that many patients, that's not feasible.
So FSGS was stuck.
A disease desperate for therapies, and an endpoint regime that made trials almost impossible to run.
So, PARASOL was formed.
A research collaboration between nephrologists, biostatisticians, patient advocates, drug companies and regulators to solve this problem.
The results of this PARASOL collaboration were that, in the middle of the Phase 3 trial, Dimerix could adjust its endpoints to the easier, more appropriate proteinuria endpoint.
This is important as we’ll see in a bit.
Why would Dimerix NOT go after accelerated approvals?
Here’s why…
(and there is a good reason for it)
Reason 1: Easier final endpoint
The results of the PARASOL collaboration showed that YES Dimerix could use the simpler endpoint, proteinuria, for the full trial.
This was later confirmed by the FDA.
HOWEVER, for the accelerated-approval endpoint, the PARASOL collaboration said that Dimerix could use the simpler proteinuria endpoint…
BUT would need to confirm with eGFR (the hard endpoint) at the end of the trial.
So, going after accelerated approval carried a greater risk that the trial would fail to meet the criteria for full approval.
Reason 2: Statistical penalty for “unblinding”
For a regulator to approve a drug, it must demonstrate that it is safe and effective at a specified level of statistical significance.
It must be more than 95% chance that the result wasn’t a ‘fluke’.
Any standard clinical trial with a single look at the end carries a 5% risk of a false positive.
This is your alpha.
Every time a trial team takes an interim look at the numbers, they create another opportunity to fluke a good result.
This is called alpha inflation.
So, to balance the scales, the trial uses what is known as the alpha spedning function.
Dimerix has a total 5% alpha to spend across both the interim and final results.
(or it risks that the trial was a fluke).
If it chooses to pursue the accelerated approval pathway, it has to allocate some of that alpha to the interim analysis and some to the final analysis.
Because each time it looks at the data, the alpha goes up.
So, Dimerix chose not to pay the statistical penalty of looking at the data twice, to give it the best chance of securing a final approval.
So… why did they not pursue accelerated approvals?
Dimerix had been working on this drug since 2012.
Hundreds of millions of dollars spent on research and development so far…
It didn’t want to risk getting to market and then having to pull the drug later.
Running a clinical trial is like playing a game of poker with a deck that you chose.
You want to put as many ‘aces’ in the deck as possible.
Make sure that the trial is properly powered.
Make sure that you have the right “endpoints”.
Make sure that you have enough ‘alpha’ to prove the trial results weren’t a fluke.
If Dimerix had chosen to pursue accelerated approvals, it could have been dealt an earlier hand, but would have had to take out a few of those aces in the process.
(particularly the statistical penalty and change in endpoints)
Now, this decision puts Dimerix in a very uncomfortable position (and it is a big reason why the stock dropped).
The market was expecting accelerated approvals, but Dimerix didn’t go down that path.
Also, now has 2 more years of clinical trials to complete, and only $38 million in the bank (as of December 2025).
The next natural question that the market is asking is, who will fund the rest of the trial?
How does DXB fund the rest?
Right now, Dimerix has $38 million in the bank, with an updated number in the quarterly report to be published in the next few days.
Now, I expect burn to decrease.
The company has completed recruitment for the trial (which is one of the most expensive line items), and sites will begin closing as the trial winds down to the last few patients.
Based on the last four annual reports, the company has burned $85 million in cash over the past four years while running the trial.
Each quarter burns between $5M - $15M.
The back-of-the-napkin maths says that the current cash balance isn’t enough to get them to an approval.
So a capital raise is expected.
Unless Dimerix can find a non-dilutive funding option, and it only has one more major region to license off - China.
The way a friend put it to me on the phone yesterday:
Dimerix either needs to raise more money on the market OR sell the China rights with enough upfront capital to get through the trial.
It’s China or Cap Raise.
IF Dimerix had gone after accelerated approval, a few of those $1.4 billion in milestone payments could have kicked into gear.
Which would have meant that the company was funded.
BUT this pathway does mean they are a bit short on capital and have limited options: China or a Cap Raise.
For what it's worth, while I traded the stock in the hope that they would get accelerated approval (and then sold my shares when they didn’t), there is always a chance for me to buy back in.
But I do want to see a solid funding strategy first.
Some of my lessons from the DXB story
Dimerix was the first ever biotech stock that I wrote about and invested in.
I followed the entire saga of its Phase 3 clinical trial for FSGS.
I look back on that article I helped write in 2021 and just think… boy, I was naive.
But Dimerix had it all, the ups (and they were big), the downs and everything in between.
For this next section, I wanted to catalogue my lessons from watching this story play out over the last 5 years.
Lesson 1: Things always cost more than you expect
I remember when Dimerix first raised capital for the Phase 3 FSGS clinical trial $20M at 20 cents.
This, according to the company, was “enough to Fully Fund the Company… through to an interim result”.
(which at the time was 72 patients).
Over the next few months, the company had a modest cash burn, working through the ethics approval, IND and ultimately the first few sites coming online.
Everything smooth.
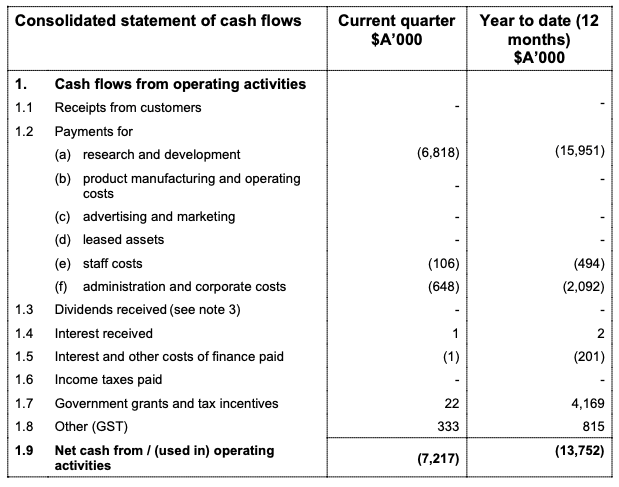
I will remember that first quarterly report after the trial results as clear as day.
$7,200,000 in outflows in one quarter.
That was almost 40% of what they had set aside to get them all the way to the interim result!
(Source, Dimerix Announcement, July 2022)
I was in Coffs Harbour at the time in this dodgy motel on a site visit to a gold mine.
I actually couldn’t believe it.
They had just raised $20 million a few months earlier… how would they have enough to get to an interim result?
And I was right… the company didn’t have enough money to reach an interim result and had to raise capital at a depressed price.
Lesson 2: When the market gives up on a stock, that’s the best time to buy
That capital raise to get Dimerix through to an interim result was in mid-2023.
Peak inflation, peak interest rate rises.
Along with the rest of the biotech industry, DXB was in the doldrums.
… and it was clear it needed to raise money.
The stock had drifted and drifted and drifted…
But that capital raise at $0.08 with a two-for-two option (at a $30 million market cap) was one of the best I saw in my time covering biotechs.
A very difficult cheque to cut. But a very good one all the same.
Because less than four months later, Dimerix signed a licencing deal with Advanz Pharma (including a €6.5 million upfront fee).
The stock doubled overnight, and the sky was clear for the interim results.
(where it doubled again, and then doubled AGAIN on a US licensing deal 18 months later).
Top to bottom, this trade was a ten-bagger for anyone brave enough to take the punt.
Lesson 3: Things always take longer than you expect
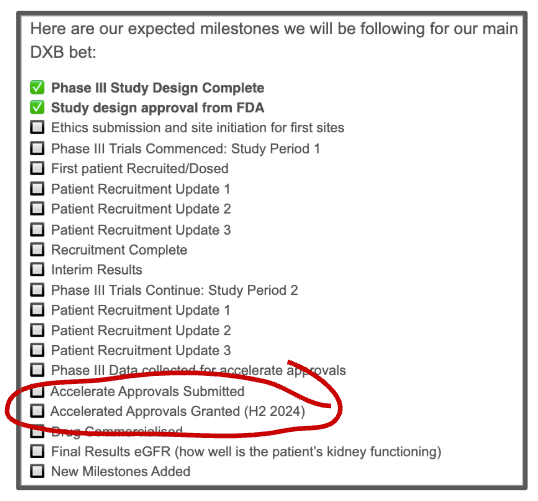
As I was going back into that first article that we wrote, I pulled up this gem.
Here were the milestones that we expected for Dimerix, including the timeframe for accelerated approvals (2024! That was ambitious):
(Source, Next Investors 2021)
At the time, the story was simple.
Orphan drug designation, recruit enough patients, and accelerated approvals.
But as the trial progressed and the PARASOL working group's outcome was published, it became clear that accelerated approvals were a hopeful ambition rather than a defined strategy.
Yesterday’s news confirms this to be true.
What I have since come to understand is that most biotechs run their clinical trials in this way…
They hope the regulatory environment will fall in their favour, BUT until they get confirmation from the FDA, it's still largely guesswork.
(and sometimes even with the FDA advice, it's still guesswork - just look at Mesoblast!)
Lesson 4: The chart doesn’t lie
A friend of mine likes to remind me of this one. The chart doesn't lie.
DXB closed at A$0.62 on 24 December 2025.
By the time today's announcement landed, the stock was already at A$0.39, down nearly 40% in just over four months.
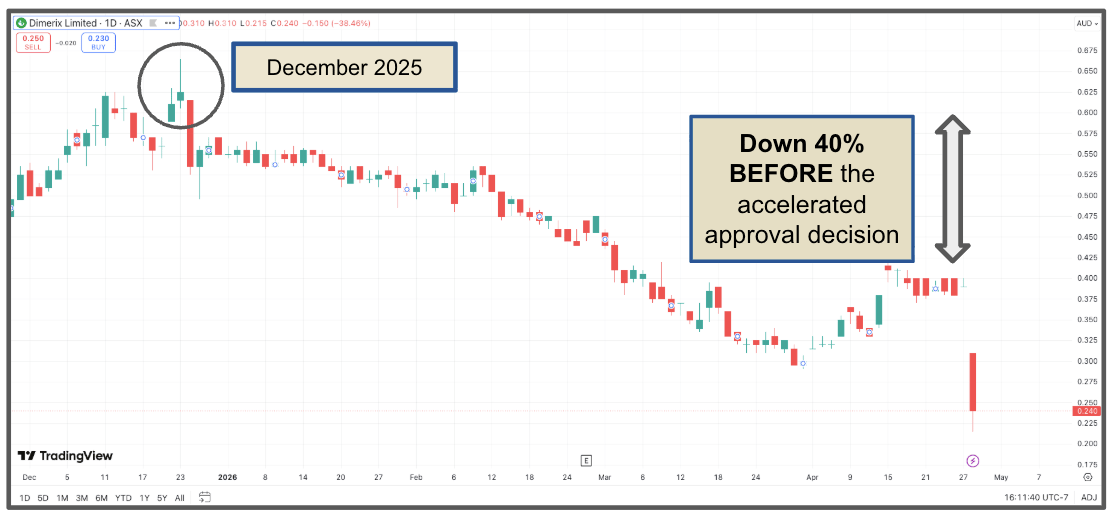
The “smart money” probably saw the writing on the wall…
Fewer mentions of “accelerated approvals” in slide decks, a clear read through on the PARASOL guidance, etc.
So, while I liked the trade, the chart was signalling that accelerated approvals were unlikely.
I'm not saying you should trade off charts. I'm saying when there's a binary catalyst pending, and the stock is grinding lower into it without a clear macro reason… listen.
Lesson 5: There is always an opportunity to get back in
This is the part I keep coming back to.
DXB has been one of the best and most painful biotech stocks I've covered.
It's also a story that isn't over. Not even close.
Here's where things actually stand today:
Trial is fully recruited (333 adult patients)
Trial is >90% powered for the proteinuria primary endpoint
Last patient last dose: March 2028
Final readout: shortly after, with potential traditional approval submission to follow
Travere has just been approved on the same endpoint, by the same regulator, in the same disease; the regulatory path is no longer hypothetical
Four commercial partners across seven major markets
~A$1.4 billion in potential upfronts and milestones still outstanding
Potential for China deal to smooth over capital requirements between now and the end of the trial
Market cap today: ~A$144 million
I will be watching the Dimerix quarterly very closely over the next few days to see just how much cash they have left.
As I’ve always said, time in the market doesn’t always beat timing the market.
But after so many years of watching this company's ups and downs, I think I’m going to want to own some stock when that final readout comes.
Thank you to Dimerix CEO Nina Webstar for all the conversations over the years and for taking time out of your day yesterday to clarify a few things for me before I wrote this article.
See you all tomorrow,
The Armchair Analyst.
The Pulse Check
Emyria (ASX: EMD) expands its psychedelic treatments for PTSD and Treatment-Resistant Depression into New South Wales. Authorised prescribers up from 6 to 12. (EMD)
🪑 National rollout very much on track, with the most interesting news to me being the increase in authorised prescribers.
Authorised prescribers are a major bottleneck for Emyria in delivering and scaling up clinics.
Doubling the AP count means that they can run more therapies at once. Each AP is responsible for multiple rooms, and each room hosting a session generates $10,000 in revenue for Emyria per day.
So these new APs effectively unlock $40,000-$50,000 in therapy capacity per day for Emyria. (assuming each clinic has 4 rooms)
APs will generally work once a week, so getting more on board is essential to running clinics at full capacity.
Emyria is capped at $40M and is my largest personal position.
TrivarX (ASX: TRI) appoints Dr. Danielle Meyrick as CEO. (TRI)
🪑 Held senior roles at Telix, nice get. Will support with clinical trials for the early-stage radiotheranostics product.
Mesoblast (ASX: MSB) achieves patient recruitment target for a pivotal Phase 3 trial in chronic low back pain. Top-line results expected by mid-2027 and potential FDA filing in Q3 2027. (MSB)
🪑 Very nice. Looking forward to these results.
AdAlta (ASX:1AD) announces its first Australian manufacturing work order with Cell Therapies for its Chinese-licensed CAR-T therapy. (1AD)
LTR Pharma (ASX: LTP) enters a trading halt pending an announcement on interim clinical trial data for its pivotal Phase II study of SPONTAN® Nasal Spray for erectile dysfunction. (LTP)
🪑 Nice. 🍿
Regis Healthcare (ASX: REG) appoints Andrew Kinkade as Managing Director & CEO. (REG)
The Report Card
Lots of quarterlies were released today. I’m just going to highlight the ones that caught my eye.
Alcidion (ASX: ALC) positive operating cash flow of $1.7M, $14.5M cash receipts, and new TCV sales of $11.7M. Full year revenue expected to be $50M with EBITDA in excess of $5M. (ALC)
🪑 Another nice quarter from Alicidion. I’ve been tracking this one closely. It's great to see positive operating cash flows.
Genetic Signatures (ASX: GSS) still had $25.7M in cash. (GSS)
🪑 Trading a bit above cash backing now. I might have missed the trade on this one
Visionflex Group (ASX: VFX) reports $1M unaudited revenue for Q3 FY26, up 24% on the pcp, and $1.99M ARR, up 20%. (VFX)
🪑 Capped at just $4.5 million, I’ve been watching VFX for a while. Looks like the company is still in that “cost saving” & “reset” mode rather than revenue-ramp-up mode.
Enlitic (ASX: ENL) reports record Q1 FY26 contract wins totaling A$5.1M, including key US$1.5M Penn Medicine and US$1.2M Parkland Health agreements. (ENL)
🪑 Really good quarter from ENL, with three major contracts.
However, cash burn remained quite high.
It will be very interesting if it can sustain the sales momentum into this next quarter, or if these deals were a flash in the pan.
Watch this space.
M&A, Big Pharma Wants a Wife
Eli Lilly is acquiring biotech startup Ajax Therapeutics for up to US$2.3 billion to develop a next-generation drug to improve JAK inhibitors used in blood cancers and immune diseases. (Biopharma Dive)