Good morning,
Welcome to today’s edition of The Armchair Analyst, a 5-minute daily update on the ASX life-sciences sector.
For about three months now, all I’ve been hearing from my wife is to make sure I eat 30 different plants each week to manage my gut microbiome.
This was the first time I heard of it…
But also, I’m a laggard when it comes to diet and health trends.
After looking into it more closely, gut microbiome has been slowly trickling into the mainstream vernacular over the last few years.
And there is one ASX-listed company at the forefront.
The next company on my Biotech 165 Challenge: Microba (ASX: MAP).
But first…
The Pulse Check
I had to check if my ASX announcement tracking feed was broken this morning…
Nope, still working.
But barely a single material announcement in sight.
CSL lands contract to supply Canada with 15 million vaccines in the event of a flu pandemic. (The Capital Brief)
This week was the UIC conference, Australia's largest medical cannabis event. The main talking point was whether the TGA would reschedule THC to a Schedule 9 drug. (Cannabiz)
🪑 This would be a big disruptor to the industry. I’ll need to unpack it more to identify the “winners” and “losers” if THC were to be up-scheduled.
But regulatory uncertainty is never good for business.
Prime Medicines has applied for accelerated approval in an ultra-rare disease category, with a two-patient gene-editing dataset. (Biopharma Dive)
🪑 Two weeks ago, the FDA released a framework for individualised therapies for ultra-rare diseases.
This will be the very first test case under the new framework.
Does this open up a whole new opportunity pathway for drug developers? Or will it be dead in the water on arrival?
Big test here.
M&A, Big Pharma Wants a Wife
Radiology firm RadNet will buy Parisian imaging and CT scanning company Gleamer for €215 million, signalling the company’s first European market entry. (Fierce Biotech)
Under the Microscope
Health trends move in cycles.
Fat-free foods.
Low-carb diets.
Organic everything.
Gluten-free.
Supplements.
Plant-based meats.
(Still not convinced on that one.)
But underneath all these trends sits the same idea:
What we put in our bodies determines how long and how healthy we live.
Over the past decade, scientists have begun looking in unexpected places for answers.
Inside our gut.
10 years ago, the “gut microbiome” wasn't a thing.
Today, we know our digestive system is home to trillions of bacteria that help break down food, regulate metabolism, and influence overall health.
My wife has been on this train for a while… and yes, we’re doing the 30-plant-a-week diet.
(She has assured me that ‘cacao beans’ in a Snickers Bar does not count).
But this trend of managing gut health has been brewing for a while.
Here is a chart of the Google Trends search for the term “gut microbiome” over the last 10 years:
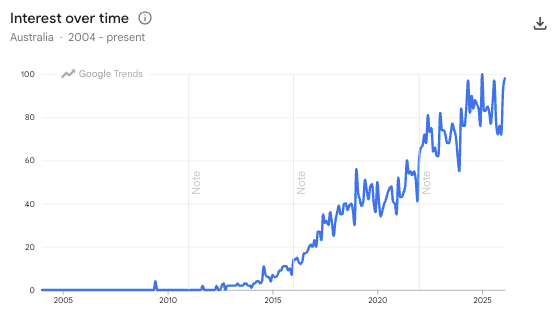
(Source, Google Trends)
The science is still relatively new, but the consumer interest is there.
If enough pilates mums are talking about “gut microbiomes” at their kids' football practice… punters, it's time to pay attention.
I am of the firm belief that retail investors make decisions macro theme first.
There is a trend or topic they see coming and want to speculate on whether the thing they saw before everyone else becomes mainstream.
But how do you bet on a trend?
Well… you find a stock that is leveraged to that outcome.
So, that's why I was immediately interested when I came across Microba (ASX: MAP) - an ASX-listed company at the forefront of the gut microbiome story.
Oh, and its largest shareholder is Sonic Healthcare.
(Only Australia’s largest pathology business)
What’s the story?
Microba (ASX: MAP) is a diagnostics company that analyses stool samples to sequence the bacteria living in your gut and provide you with insight into your gut microbiome.
There is a bit more to it than that, but that’s the gist of the product.

In anticipation of a meeting with the CEO this Monday, I did some field research.
Basically, I went to my health food store to get my gut tested.
Turns out, the product that Microba sells is NOT direct-to-consumer, but sold through naturopaths, physicians, or doctors who can refer you to the test.
Lucky for me, there was a naturopath at the store who could answer my questions.
(thanks Felicity!)
What she told me was that Microbiome Explorer is like the Rolls-Royce of gut microbiome testing.
It’s expensive, but very good.
If someone was, say, gut-microbiome-curious, where they just wanted to get an idea of what was going on in their gut, she would likely recommend a cheaper alternative.
A basic blood panel, for instance.
BUT, if there's a serious underlying issue, IBS, or something similar, then Microba’s product was the test that provided the most comprehensive evaluation available.
This was a very deliberate call by the Microba team.
They wanted to position themselves as a premium medical diagnostics product for gut issues, not a direct-to-consumer product.
The market is targeted, and you can charge more…
But it is also a slower and harder mountain to climb.
(As I've seen with many diagnostics companies, adoption is the hardest bit.)
Quick word on the numbers
Over the last few years, MAP has been winding down sales from legacy products to focus on growth in its core testing business.
This has meant topline revenue has been a relatively slow grind, sitting stable at around $7M every six months.
The company is still operating at a loss, but MAP is targeting regional breakeven in Australia and the UK by the end of FY26.
Regional breakeven is a fancy way of saying the breakeven for the business's sales vertical, excluding therapeutics R&D and corporate costs.
(So, not really breakeven)
With the legacy products now fully wound down, the next half-yearly results will be very revealing.
Here is the chart showing Microba’s topline revenue year-on-year.
(If you added it all together, it would look flat, but it's important to break down the growth by vertical)
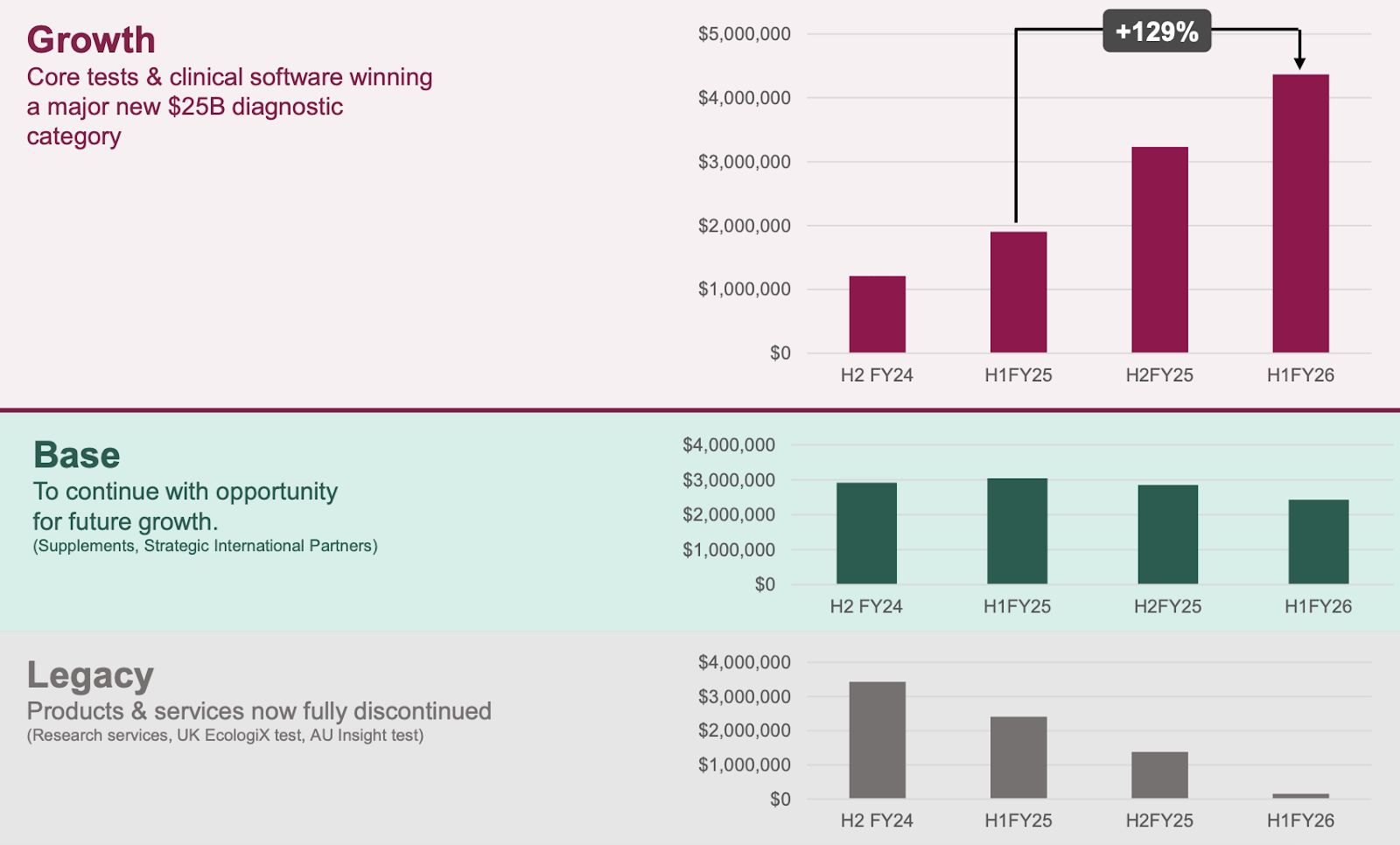
(Source, MAP quarterly report page 8)
The next half will be the first time the legacy products no longer contribute to topline revenue. So all growth (and business attention) should be focused on the core testing platforms.
Microba hasn’t yet reached “sustainable business” status.
And is burning around ~$4 million each quarter, which means that it will be back to the market hat in hand within the next 12 months.
Unless… it can sign a deal on its therapeutic asset.
(and I’ll let you know why I think that might be a hard ask)
Microba’s therapeutic blue sky punt
Microba has invested time and capital in developing its lead asset, MAP 315, for inflammatory bowel diseases.
It has taken it through to a Phase 1, with a Phase 2 IND submission being compiled.
(so that it can run trials in the US)
It has developed a Live Bioteherapeutic Product (LBP)…
Think of this like a probiotic, but with extra regulatory steps and protections.
Probiotic = Yoghurt isle (FMCG commodity)
Live Biotherapeutic = Doctor’s prescription (Medicine)
To develop the asset, Microba has used all of the data from its tests to identify which LBP work best for which indications.
That's essentially their R&D edge.
There have been a couple of big transactions in the gut health therapeutics space (the biggest being Prometheus's US$10.8 billion acquisition), which validates the opportunity.
But Microba currently has no plans to advance its therapeutic assets beyond Phase 1 without a partner first.
There's a world where Microba outlicences these with an upfront fee, reducing strain on the balance sheet.
And there is a nice slide in the last quarterly presentation that shows all the M&A transactions.
My take, though…
They'll likely need to develop through to a Phase 2 before this becomes genuinely attractive to a big pharma buyer.
It’s not out of the realm of possibility to partner at just a Phase 1, but I would expect it to be a lot easier if they had some human efficacy data first.
Now, of course, developing through to a Phase 2 costs money, so the company has (rightly in my opinion) chosen to allocate precious capital directly to its diagnostics business - the core driver of growth.
So when I’m evaluating Microba’s therapeutics potential, I’m giving it a very low chance of generating any upfront value without the company investing more in its development.
But, if they do manage to secure a licensing deal…
It's a surprise to the upside.
How does Microba fit the “Armchair Framework” for diagnostics products?
Last week, I revealed the four pillars of any diagnostics business.
Approvals: just get you to the starting line, but they are necessary to bring the product to market.
Logistics: all the unsexy work of collecting samples, ensuring they are properly stored, and managing and maintaining records.
Adoption: Will people use it? Will doctors prescribe it? This requires extensive education and thought leadership from key opinion leaders in the field.
Finally, Reimbursement (by the government or insurance companies). Get paid.

Underpinning it all is clinical evidence, which is built up over time.
So, let’s see how Microba stacks up:
Approvals
Microba’s flagship product, Microbiome Explorer, is approved for sale in Australia and the UK as a Lab Developed Test.
Logistics
Sonic Healthcare is the key distribution partner in both Australia and the UK - and, as mentioned, is also the top shareholder.
The test is stool-based and is prescribed by a naturopath or physician.
Not a direct-to-consumer product, which is a deliberate choice.
Adoption
Now, this is where it gets interesting.
The key drivers of adoption are clinical evidence, key opinion leaders, and extensive lobbying.
(basically glorified industry sheep herders)
What I liked about Microba's strategy is that there's a very clear outline for how they plan to drive adoption. And they've only just scratched the surface.
Think of the classic technology adoption curve.
The innovators - like Felicity, our friendly naturopath - are quick to embrace this product.
But the late majority, the crusty GPs who've been doing things the same way for 30 years, will take much longer.
Microba is still at the very early stages of the adoption curve.
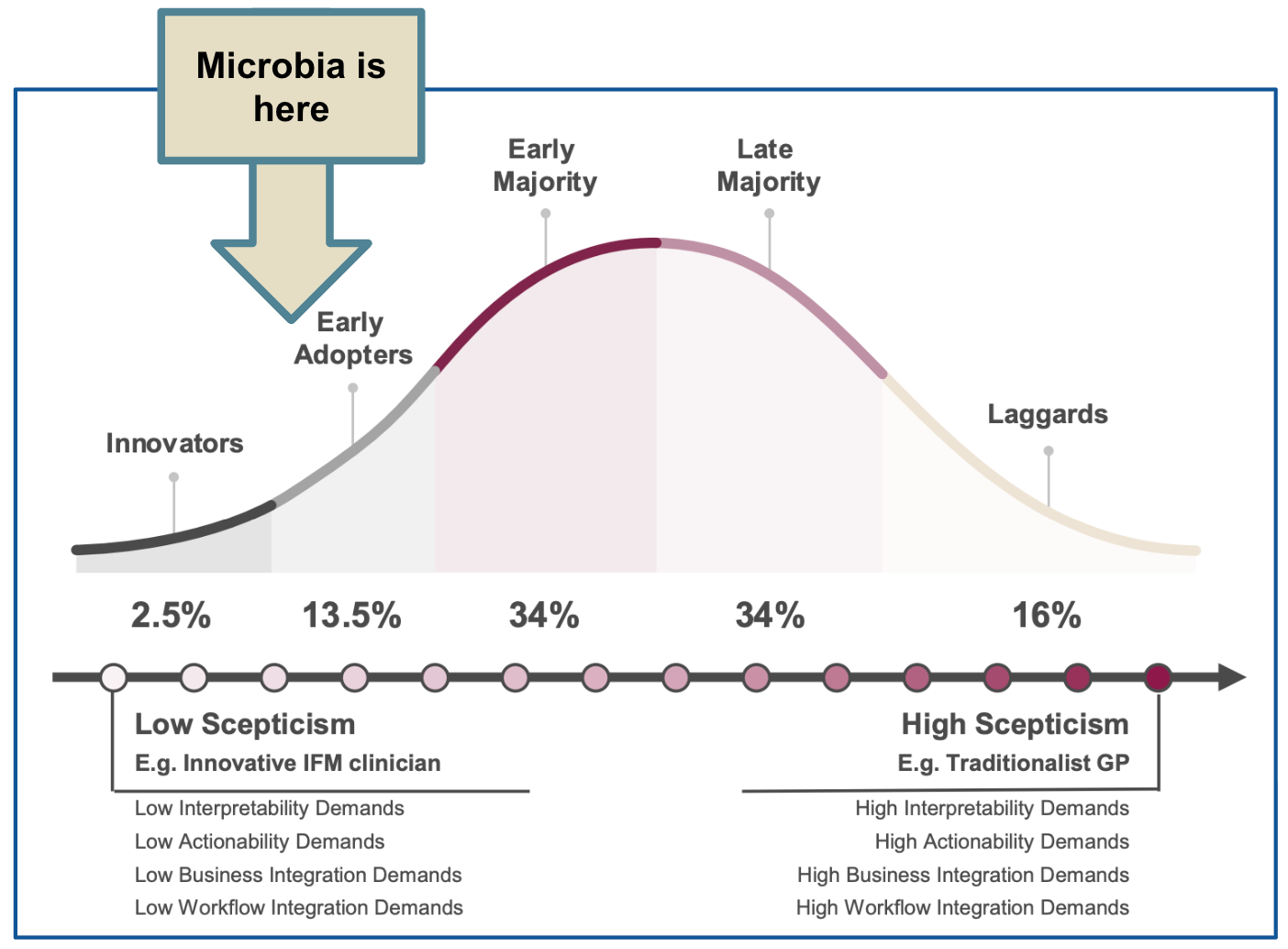
Where many diagnostic companies fall over is that they try to parachute into the middle of the technology adoption curve.
Selling to the Late Majority and relying on reimbursement as the primary adoption driver.
Microba is different.
It is not relying on reimbursement as the primary driver of adoption.
Reiumbursement
Microba does not have a reimbursement strategy in place.
Speaking to CEO Luke, Microba wanted to build a product that sells itself first, with reimbursement as an unexpected upside much later.
So, double down on adoption. Don’t wait for reimbursement to do the heavy lifting.
It's an unconventional strategy for a diagnostics company. But given how hard reimbursement is to obtain (and how long it takes), there's logic in it.
So, what’s the bet here?
In the long term, this is a macro-driven bet.
It is a bet that the gut microbiome industry will continue to grow into the mainstream, and that Microba will be at the forefront of testing when it does.
The market is large. The product is premium. The distribution partner is credible.
Microba just need to get the market education piece right and navigate the ‘come raise’ game that every publicly listed company plays, as they continue to grow and scale the business.
A big thank you to CEO Luke Reid and CFO James Heath for walking me through the Microba story.
See you all tomorrow,
The Armchair Analyst





