Good morning,
Welcome to today’s edition of The Armchair Analyst, a 5-minute daily update on the ASX life-sciences sector.
Alright, today is crunch day.
In December last year, I took a trading position in Immutep (ASX: IMM) after it announced a US$370 million licencing and commercialisation deal with Dr. Reddy for its Phase 3 cancer asset.
Today, Immutep announce the results of its futility analysis.
Either the data is good enough to finish the trial, OR its back to the drawing board.
A true boom or bust moment.
I only wrote one version of this article last night. (Please don’t fail!)
UPDATE: We may not get a result today, as the stock has just been suspended.
Is this good or bad? Who knows.
… delays always make the market a bit nervous.
But first…
The Pulse Check
Intelicare Holdings (ASX: ICR) has requested a trading halt pending the announcement of a “material strategic partnership and long-term commercial agreement”.
🪑 This could be good 🍿
Rhythm Biosciences (ASX: RHY) first commercial sale of its colorectal cancer diagnostic test. (RHY)
Patrys (ASX: PAB) initiates manufacturing for its injectable formulation for delirium. Pre-clinical trial scheduled for H2 2026. (PAB)
4DMedical (ASX: 4DX) is added to the All Ordinaries Index in the March 2026 quarterly rebalance. (4DX)
🪑 More passive buying from passive Index funds is good, HOWEVER, there will be more borrow available for shorting positions.
Have a look at the Christmas tree chart from Clarity Pharmaceuticals (ASX: CU6) in 2024 to see how some of these stocks can trade when they make it to the Index.
Good in the short term; will see how it plays out long-term.
Adherium (ASX: ADR) generated $198,000 in sales for its smart inhaler monitoring product in January and February, with 3,071 RPM devices shipped and 2,183 activated. (ADR)
🪑 The question I’d be asking: Is Adherium on track to reach breakeven of 6,000 activated patients by June this year?
Based on these numbers, it is on track from a “devices shipped” perspective, but slightly behind on “activations”.
That said, these numbers are pretty strong, and they could still get there by June. But it will take a mighty effort.
Adherium (ASX: ADR) also presented health economic and behavioural data from a study with Intermountain Health, showing cost savings and improvements in patient health from using the ADR smart inhaler device. (ADR)
🪑 This data will be important for phase 2 of Adherium's commercialisation strategy: Value-Based Contracts.
I wrote about ADR a couple of weeks back, worth a read: The Price of Buying Time: The Adherium (ASX: ADR) Story
Last week, a surgeon in London removed a patient’s prostate while the patient lay in a hospital in Gibraltar 2.4 km away. The entire operation ran with just a 60-millisecond delay, and it worked flawlessly. (The Guardian)
🪑Telesurgery is here. Technology is awesome.

Cash Injection
EVE Health Group (ASX: EVE) is in a trading halt for a capital raise. (EVE)
🪑 I thought there was something odd about the announcement yesterday… all makes sense now.
When you see these long, update announcements that don’t really mean that much but have a bunch of progress information, they are what I like to call “cleansers”.
They effectively cleanse the market of all relevant information, leaving a clear sky for a capital raise.
It’s a big tell.
I’ll make sure to point one out next time I see it.
M&A, Big Pharma Wants a Wife
Universal Health Services buys Talkspace for US$835 million. (Second Opinion)
🪑Some good coverage of the deal by Second Opinion, including how this sets off a ripple of projected 2026 digital health acquisitions.
Under the Microscope
Bet’s placed.
The roulette wheel is spinning.
No numbers. Just red or black.
Either the trial works, or it doesn’t.
Today, I’m covering Immutep (ASX: IMM), a cancer drug developer that is in the middle of a Phase 3 cancer combination trial in first-line non-small cell lung cancer.
(The “first line” is the first treatment a patient gets for cancer; it is the biggest and most valuable market.)
378 patients globally, with 50% of the trial target enrolled so far.
(Full enrollment scheduled for Q3 CY2026)
It all comes down to this…
The futility analysis results.
A futility analysis is like an early sneak peek at the data.
An independent body will unblind the data and, based on how good it is, determine whether there is any realistic chance this trial will succeed.
If YES, the trial continues.
If NO, the trial ends early.
Now, there is a “Mega Bull Case” scenario that occurs very, very, very rarely, in which the trial may stop early due to overwhelming efficacy.
(I’m not expecting this to happen for Immutep, but a man can dream).
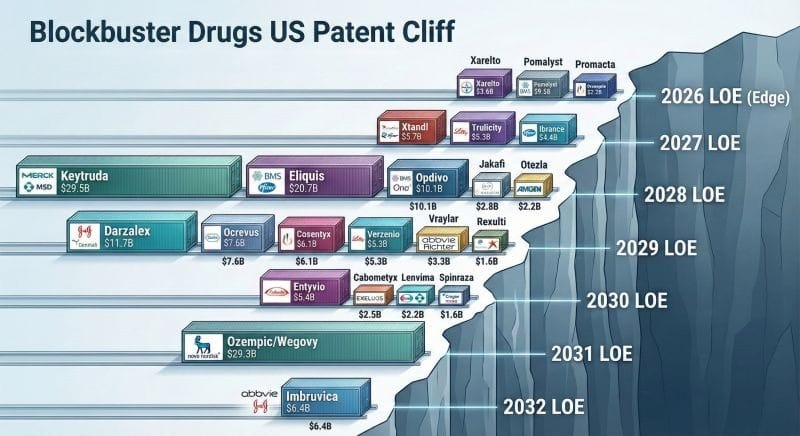
Why Immutep is in “pole position” to solve Big Pharma’s “patent cliff” problem
Immutep has developed a combination therapy for non-small cell lung cancer with Merck’s KEYTRUDA.
Keytruda is one of the biggest cash cows, generating US$29 billion in annual sales.
… the problem?
Keytruda’s patent expires in 2028.
The owner, Merck, is desperately looking for ways to extend the value of this mega-blockbuster drug.
It has diversified into several indications (COPD, IBS), with three $10 billion+ deals in the last few years.
… but it also has an eye squarely on oncology.
Last month, Merck announced that it would be splitting into separate oncology and non-oncology divisions in preparation for Keytruda coming off patent:
If these results are good, Immutep is in pole position to step in and save Merck (or any of the other big pharma companies) from the impending patent cliff with a promising new asset.
What’s the patent cliff?
When a drug comes to market, it is given a period of “exclusivity” during which no other biosimilar drugs can enter the market and compete.
This is big money for big pharma.
When drugs lose exclusivity, it paves the way for cheaper generic alternatives, leading to an 80% reduction in market share within the first year.
So all of those juice revenue dollars that drive shareholder returns for Big Pharma evaporate…
Just, like, that.
It takes ~10 years to bring a drug from discovery to market, so the fastest way for big pharma to replenish its revenue pipeline is to acquire.
(Generally, companies at Phase 2 or Phase 3 of development).
That’s why 2025 was one of the biggest years for M&A in a long time, and why 2026 is set to be even bigger.
Here is the patent cliff for some of the big drugs over the next few years.
Biggest of them all?
Keytruda…
Now, Immutep already has one licensing deal up its sleeve.
In early December, it signed a US$370 million deal with generics manufacturer Dr Reddy for everywhere except US, Europe, Japan and China.
Picking Dr. Reddy as a partner was very, very interesting.
It is not one of the household Big Pharma names, but it still has a US$12 billion market cap and is a lead distributor of generic drugs.
Dr. Reddy is currently in the process of developing its own competitor to Keytruda…
So if Merck wants to retain the license to Immutep’s drug in other key markets like Europe and the US…
It may need to move fast on a licensing deal before it gets pipped at the post by someone else.
This Dr. Reddy deal, along with positive results from the futility analysis, should put significant competitive tension on other companies looking to licence Immutep’s technology.
So the Dr. Reddy deal sets the stage, but the futility results are the spark for more deals to come.
In my opinion, there is no other company on the ASX more leveraged to an M&A transaction and to the “looming patent cliff” story than Immutep.
It’s been a long journey for shareholders.
(10+ years so far).
But it looks like all of that hard work is finally coming to a head.
So, how will I play the trade?
(Not financial advice, just what I’m doing)
IF the results are GOOD, I may look to take some chips off the table if the stock really runs, but I still want some leverage for the full Phase 3 results.
IF the results are BAD, I may look to close out the bet and book the loss.
The stock is in a suspension, so we may need to wait till later this afternoon or even tomorrow before we get the results.
Good luck!
See you all tomorrow,
The Armchair Analyst.







