The first-ever Armchair Pick is…
Tetratherix (ASX: TTX)
(Market Cap $208M | Cash Balance: $21.5M)
Disclosure: Armchair Analyst Media Pty Ltd owns 16,250 escrowed TTX shares. TTX has also engaged Armchair Analyst for investor awareness services.
This information is general in nature and does not constitute personal financial advice.
Good morning!
Since I started this newsletter, I’ve been begging for stock on the ASX to put their hand up and say:
“We are going after the massive GLP-1 market.”
So far, crickets.
Until today.
My first-ever Armchair Pick, Tetratherix (ASX: TTX), has just revealed the outcome of a five-year “stealth project”...
A nasal delivery product for longevity medicines, peptide therapies and GLP-1s.
GLP-1s are weight-loss drugs like Ozempic and Wegovy.
(heard of them?)
TTX’s nasal delivery technology is one of several platform applications that it has developed.
It’s newest, and potentially most interesting.
This is because TTX could have a product that delivers GLP-1s without an injection in the market soon.
How will it sell its product?
Through the 503(a) regulatory pathway for “combination drugs”.
Take a proven drug (like Ozempic) and a novel delivery (like TTX nasal delivery), and it is saleable through a combination pharmacy.
The catch is that combinations can only be sold through online distribution (not doctors), and there needs to be a real clinical rationale for why the person can’t take the standard medication.
(Like needle phobia)
In the US, there are a bunch of these combination pharmacies popping up…
GoodRx, Hims, Ro.
… OR, TTX’s new partner Superpower Health.
TTX just signed up to US$30 million (over 10 years) deal with Superpower Health to grant exclusive access to its nasal drug delivery technology in the US and future R&D efforts.

(Source, TTX Announcement)
The founder, Dr Ali Fathi, and the CEO, Will Knox, will host an investor webinar tomorrow, Tuesday, 17th March, at 9:00 am AEST to explain the deal:
GLP-1s are what made the first-ever $1 trillion big pharma company, Eli Lilly.
And last year, Pfizer acquired GLP-1 company Metsera for US$10 billion.
The weight-loss market is a gold mine for big pharma, and TTX will sell the picks and shovels via its novel delivery mechanism.
TTX’s vision for this product is not just to be a delivery mechanism for GLP-1s…
(and I’ll explain later in this note why that is extremely important)
… but also for a bunch of other precision medicine and fragile molecules that are hard to get into the body.
Like peptides.
(Sorry, Essendon fans, peptides are back.)
Peptides are biological signalling molecules that instruct the body to perform specific functions.
Repair tissue, regulate inflammation, stimulate collagen production, optimise sleep, and accelerate recovery…
Voices like Andrew Huberman, Peter Attia and Bryan Johnson have popularised these ‘grey area’ medications through massive platforms.
Terms like “longevity medication” and “biohacking” are now becoming mainstream.
Here is the Google search trends for peptides over the last five years:
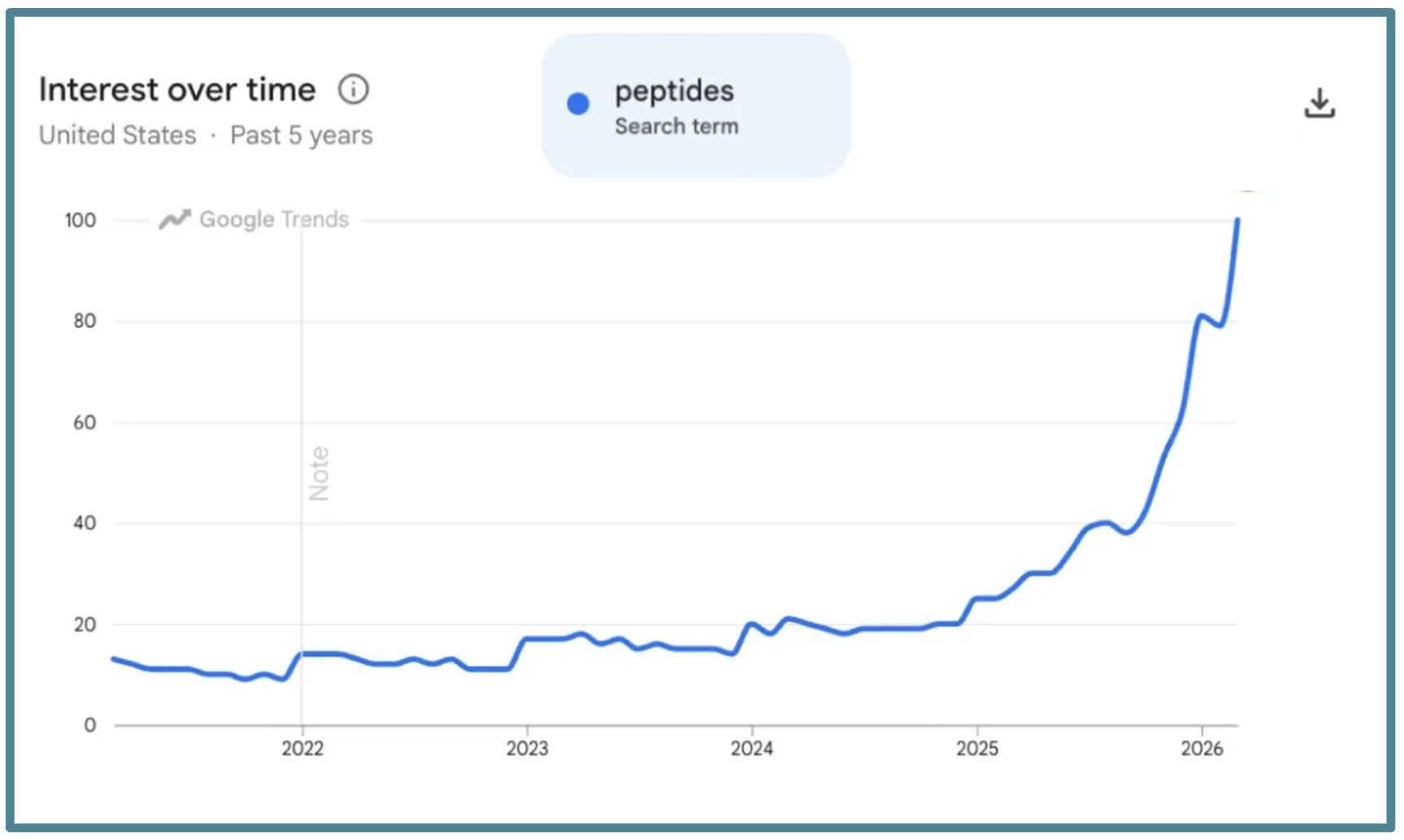
(Source, Google Trends)
RFK Junior teased on the Joe Rogan podcast last week that the US would deregulate a bunch of peptide products popular with younger generations.
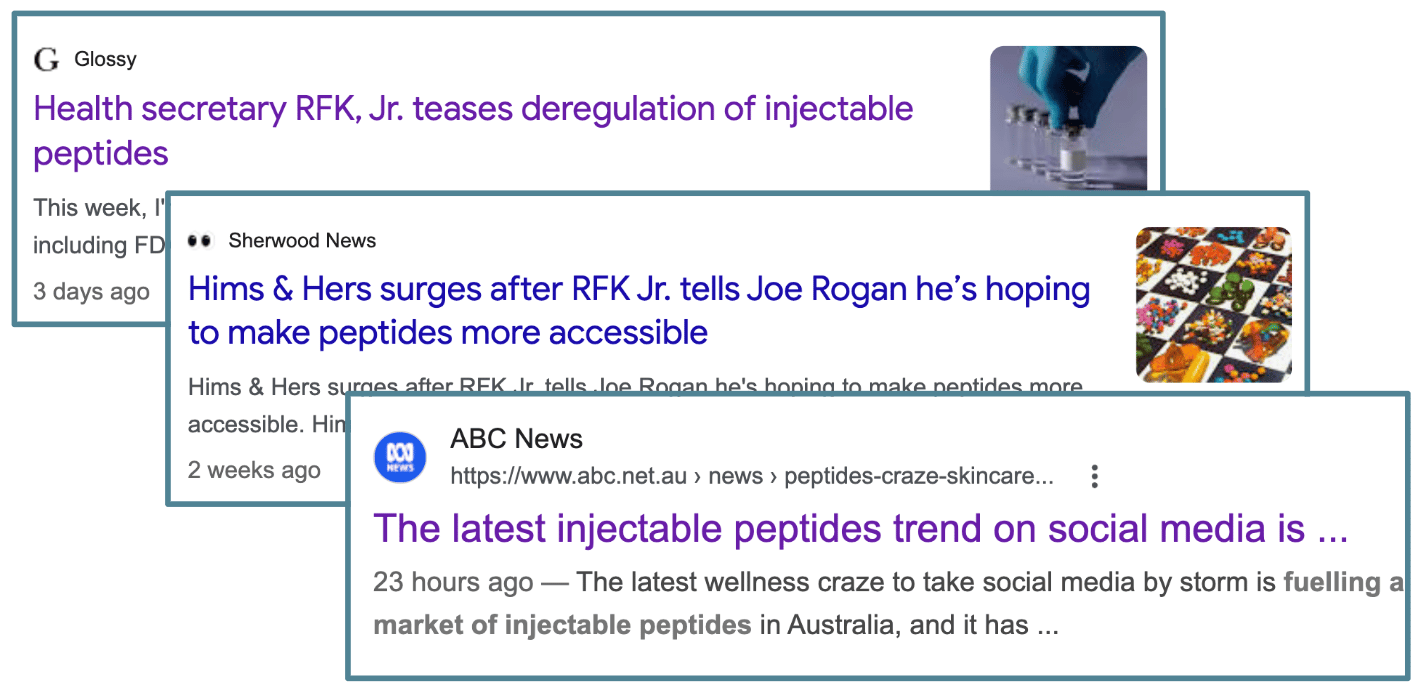
(Source, Google News)
So peptides are coming - and TTX has a much better delivery mechanism than injections.
So, what of TTX’s new partner Superpower Health?
Remember last week, that massive transaction in the Australian VC Landscape?
Eucalyptus, $1.6 billion acquisition by Hims & Hers…

(Source, Financial Times)
Eucalyptus is very similar to the company TTX just signed a deal with.
Telehealth for precision medicines.
So, with its 5-year stealth project now revealed, TTX has put itself in the very centre of two of the biggest health trends of the last few years:
Precision medicine (peptides) and GLP-1s.
But that is only just a fraction of the TTX story.
Let’s dive in and find out why I invested…
What’s the TTX story?
Early-stage biotechs love to throw around the phrase “platform technology”.
But Tetratherix (ASX: TTX) is founded on the principle of “platform first”.
A software business model applied to a medical device company.
(Makes sense that the CEO of Zip Pay is on the board and that the founder of Xero is the third-largest shareholder)
Platform technologies work because the heavy lifting happens upfront.
Building the core technology first and then scaling it across multiple products.
TTX’s technology is an injectable biomaterial that provides tissue with a surface to grow on or space to heal.
It is liquid at 25 degrees and hardens at 35-37 degrees in the human body.
This makes it a perfect technology for a range of surgical applications (spacing, bone grafting, healing).
… and now nasal drug delivery.


It’s modular, has mechanical properties similar to natural tissue, and is safe in the body.
For software companies, a platform business model works because it is scalable.
For biotechs, it is much harder due to the regulatory burden of bringing each new product to market…
But TTX’s strategy is to find a strategic partner in each target to bring the product to market.
Here are the six products that TTX is developing:
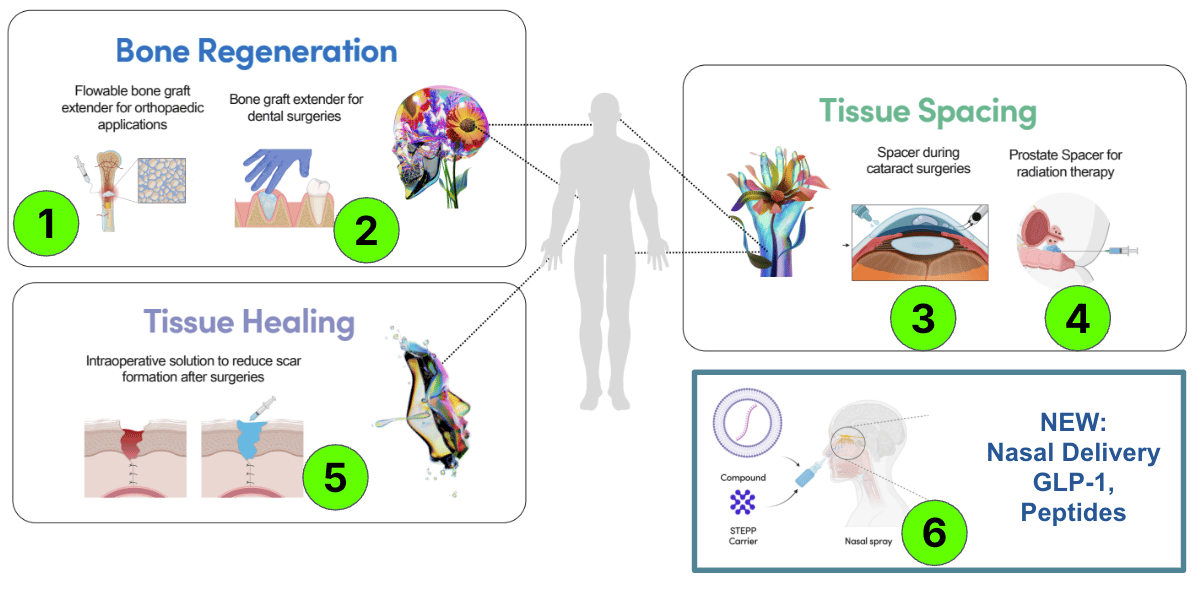
Each product is an independent “company maker”.
What ties it all together?
The platform.
So, what’s the bet?
TTX commercialises its product across multiple indications, with each shot on goal independently leading to a potential giant exit.
This is what I hope happens. No guarantees, and there are a bunch of risks and challenges that TTX will need to work through before it gets there.
My initial entry price for TTX is $4.00.
To summarise all of my thoughts, I’ve put together my investment memo for the company that highlights:
The Explainer
The 7 reasons I like the stock
The “Bet”
The Risks
The Armchair Take
Feel free to reference this memo at any point in time. Downloadable link here:

What’s TTX’s commercial strategy?
One of the big things I’ve learned on my journey investing in medtech companies is that success or failure really boils down to one thing…
Commercialisation.
Regulatory approval just gets a company to the starting gate; the real determiner of success or failure is the commercial strategy.
There are two different strategies to bring a medical device to market:
Build an in-house sales force
Partner with a distributor
The first is expensive, but it gives you more control.
The second is more scalable (particularly for a smaller business like TTX), but the risk boils down to the partner selection and execution.
Tomorrow, I’ve lined up a guest post from someone who has built sales teams in the US for multiple medtech companies before
A little teaser…
Companies should only pursue the distributor pathway if it is part of their strategy, not just to save cash.
This was the MAIN thing that I wanted to figure out with TTX before I chose to invest in the company and crown it as my first Armchair Pick.
How much emphasis did TTX put on its go-to-market strategy?
The answer… a lot.
Speaking to the CEO Will, he spent nearly four years doing due diligence on Henry Shein to select them as a distribution partner for TTX’s bone grafting product for dentists.
(Henry Shein is one of the top sellers of dental products in the world)
12-hour days in hospitals in Texas, California and other states around America, just watching how the Henry Shein reps sell their products.
Reps will only push products that actually sell.
Products that…
Solve a real problem for patients
Are fast and easy to use
Available and accessible
and then… cost.
They won’t push a product just because the science is good.
Each of TTX’s platforms is specifically designed with the end user in mind.
Fast, easy to use, and solves a real problem.
(Exactly what surgeons want)
Here is how TTX’s product comes packaged. All ready to go.
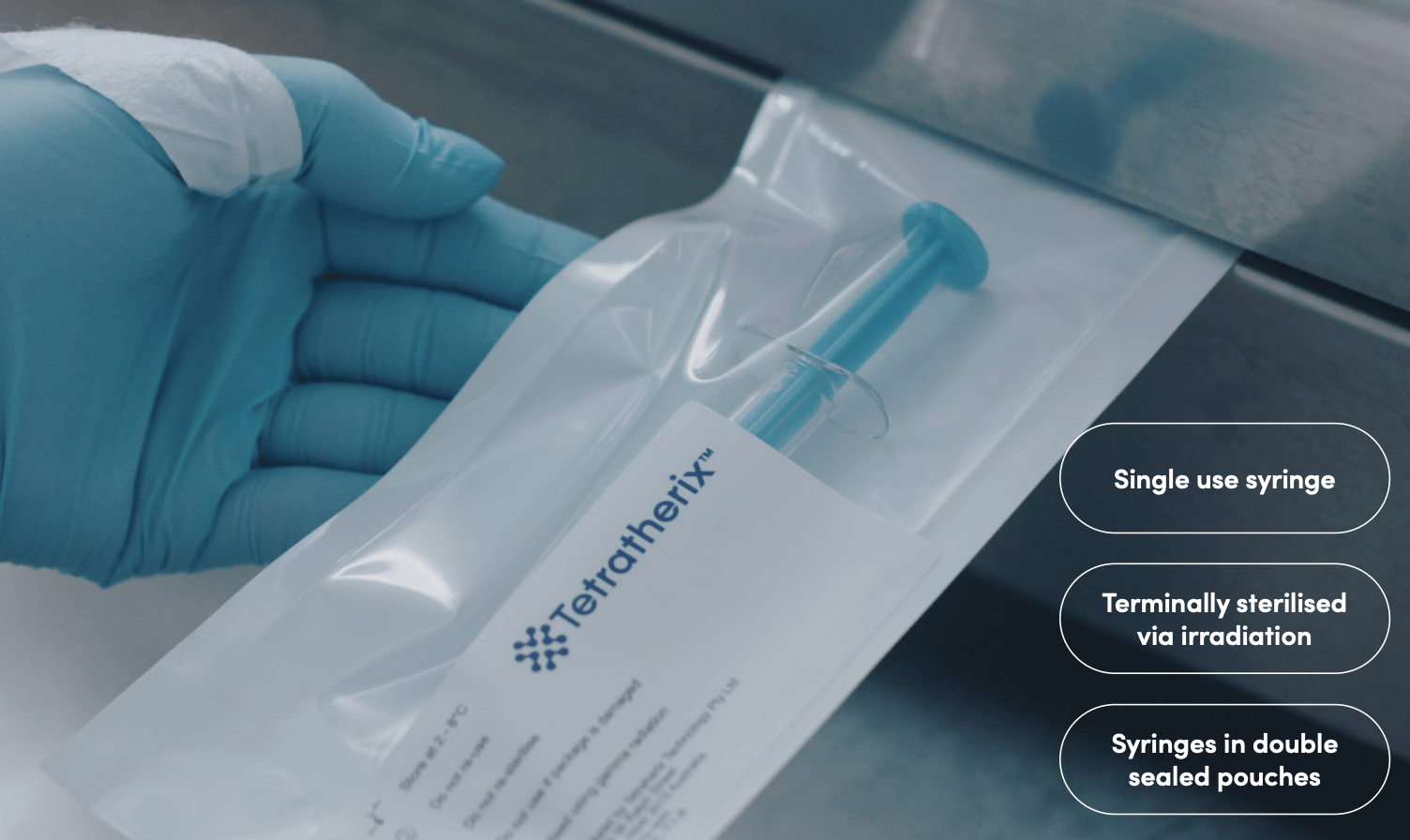
No mixing; no shining a light to harden the gel.
Just inject the fluid where it needs to go and let the body’s temperature harden the mixture in place.
TTX’s strategy is to find go-to-market partners such as Henry Schein, Alcon, and Superpower and sell them the manufactured product.
With the partners doing the heavy lifting on the sales and commercialisation front.
But execution and partner selection are everything.
The 7 reasons I invested in TTX:
The People: The CEO Will Knox has over 10 years of experience selling medical devices. For Will, “due diligence” means getting his hands dirty. He would spend 12-hour days in hospitals across America watching his partner reps sell. I like this dedication and detail.
Platform is not a feature; it's the business: One unique technology, multiple different applications. The heavy lifting is done up front, and every new product that follows is cheaper to build with a proven safety profile.
First on the ASX to leverage the GLP-1 story: I've been waiting for an ASX company to put their hand up on GLP-1s. TTX just did.
Defined go-to-market with tier-1 partners: Henry Schein for dental, Alcon for eyes. Most medtech companies fall over at the commercialisation stage. TTX designed its products around its partners.
Multiple shots on goal, funded by JV partners: The R&D costs aren't all on TTX's balance sheet. Others are paying to play.
US$30 million deal before a product even launched: TTX signed a US$30 million deal (over 10 years) with Superpower Health to sell precision medicine delivery.
Newsflow, newsflow, newsflow: Multiple FDA clearances expected this year alone.
Why I’m so interested in the nasal delivery technology
If you look at the history of Novo Nordisk and the development of Ozempic, you quickly realise something…
In drug development, small improvements in delivery can make an enormous difference.
(There is a great podcast on the history of Novo Nordisk and GLP-1s here)
First-generation GLP-1s have been around since the late 2000s, originally developed for diabetes control.
But a once-per-day injection meant they were never going to be a serious mass-market weight-loss product.
Then Ozempic changed everything.
In 2017, the next-generation drug moved from once per day to once per week. Suddenly, it was palatable. Suddenly, it was a phenomenon.
The next iteration? A pill. Locked up by the two big companies, Novo Nordisk and Eli Lilly.
But the pills have issues too…
It’s not easy getting a fragile drug like a GLP-1 through the stomach.
Gastrointestinal issues, nausea, vomiting, diarrhea, constipation, and abdominal pain.
But TTX is taking a different swing entirely.
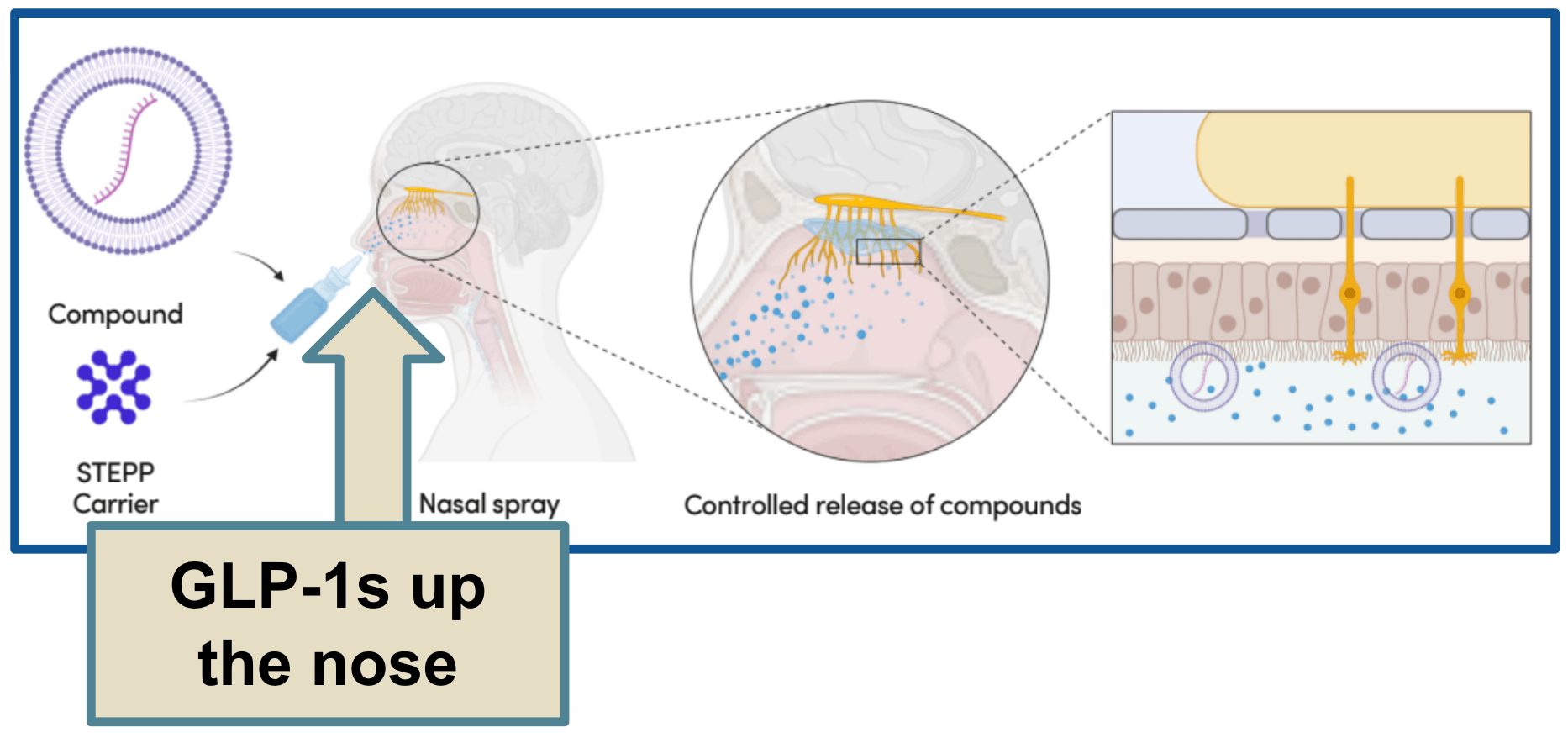
A nasal delivery product.
Now, the nose actually has excellent access to the bloodstream, the problem is that most nasal sprays just fall straight out. They never get the chance to absorb properly.
TTX's product is different.
It adheres to the nasal cavity, allowing for the slow, controlled release of drug products, such as GLP-1s, directly into the bloodstream.
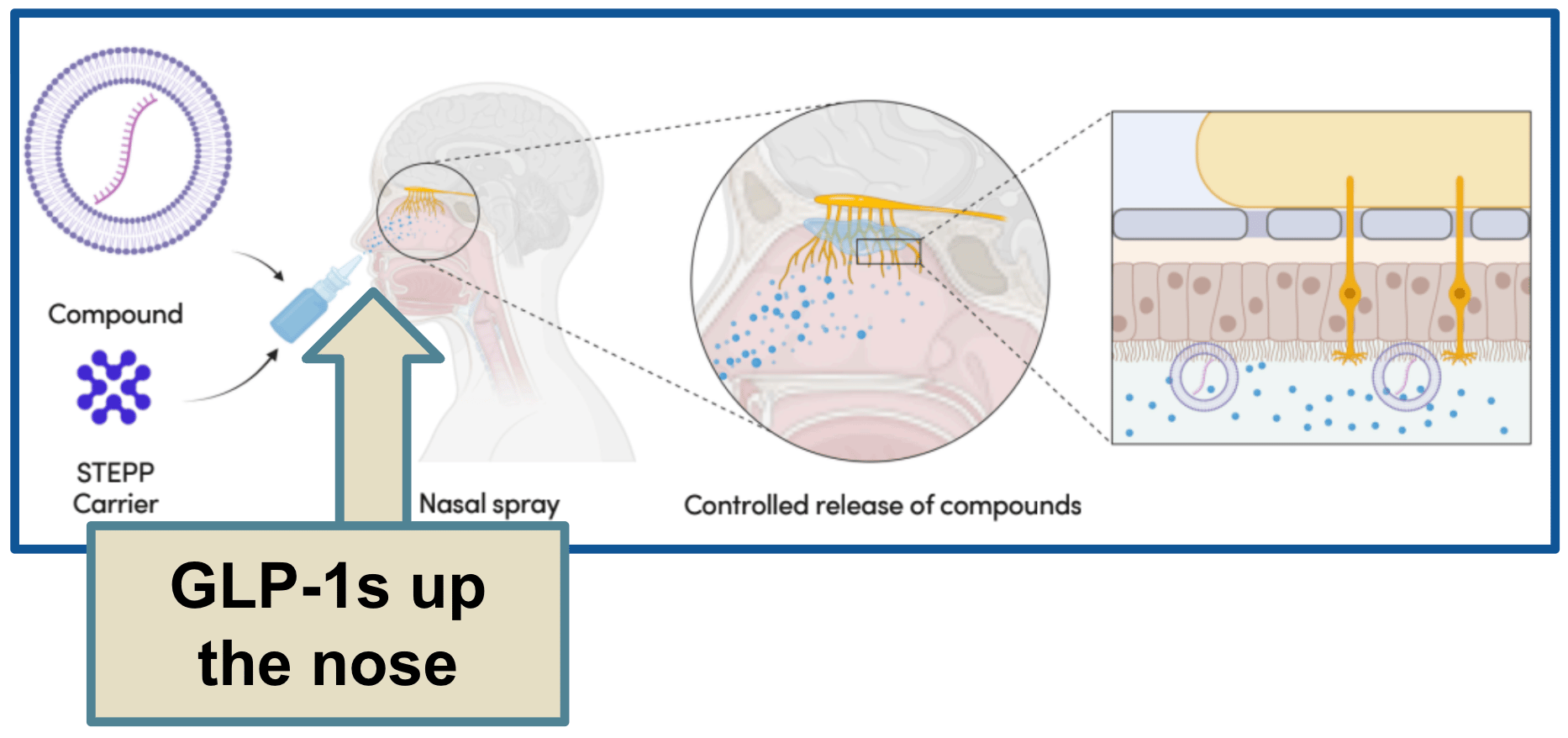
But getting it to market?
Here's where the 503(a) pathway comes in.
Under 503(a), compounding pharmacies can bring combination products to market, existing drugs paired with novel delivery mechanisms, without needing to run full clinical trials to prove safety and efficacy.
Think of the platforms already operating in this space: GoodRx, Hims, Ro.
And now, TTX's new partner: Superpower Health.
There are risks. The FDA has started cracking down on compounding pharmacies selling GLP-1 therapies combined with things like Vitamin B or levocarnitine. The regulatory environment is tightening.
But a truly novel delivery mechanism, one that solves needle phobia, with nothing else like it on the market, is a different conversation.
(If you want a deep dive into what’s happening, read: Compound weight-loss drugs are everywhere. The FDA is cracking down)
And the vision here goes well beyond GLP-1s.
Superpower and TTX intend for this platform to work across a range of molecules, peptides, fragile biologics, and any substance that benefits from improved delivery into the body.
So yes, it's a big swing.
But the prize? Even bigger.
So, what else is TTX working on?
A deep dive into TTX’s product suite
In this section, I want to take a closer look at each of TTX’s commercial opportunities individually.
What are the prospects of success, and what are the chances that it will get there?
As each of these products are sold to surgeons, I’ve enlisted the help of a friend, Surgeon Dan, who has over seven years of experience in the field and has used many of the products that TTX intends to displace.
Here is a high-level breakdown:
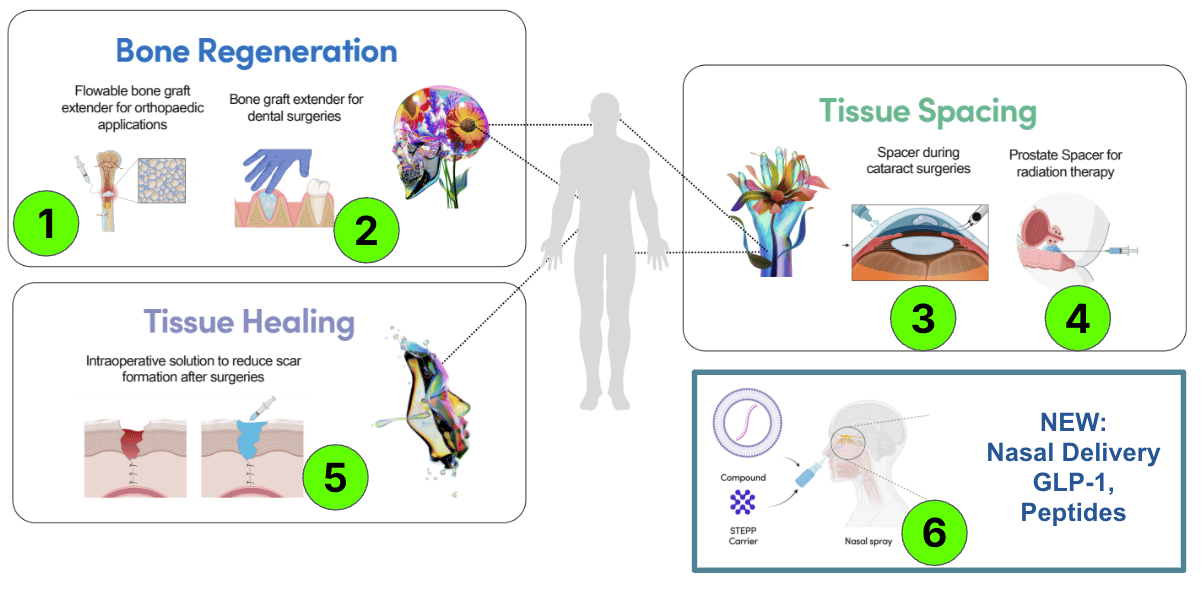
Bone Regeneration: Dentistry
What’s the product?
A synthetic, injectable scaffold that mixes with bone grafting for use in dental surgeries.
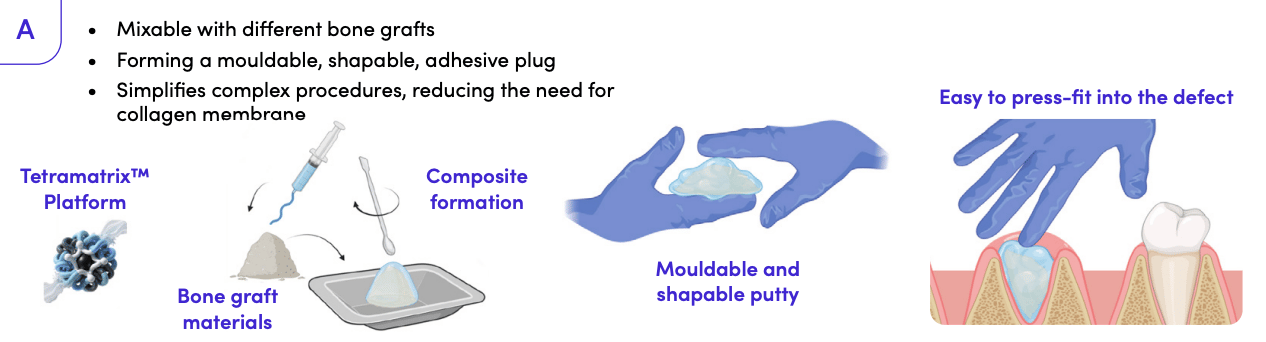
Who’s the distribution partner?
Henry Schein, largest provider of dental products in the world.
What stage is it at?
510(k) application is in, approvals this year.
What problem is it solving?
Right now, Henry Schein sells bone graft materials (BGM) for dental surgeries.
But existing products are complicated to use, so general dentists can’t perform the procedures themselves.
TTX’s product makes every single BGM better, giving an incentive for Henry Schein reps to sell it alongside BGM products - particularly to general dentists who can now do these complex procedures.
A product that makes a rep more money will be one they want to sell.
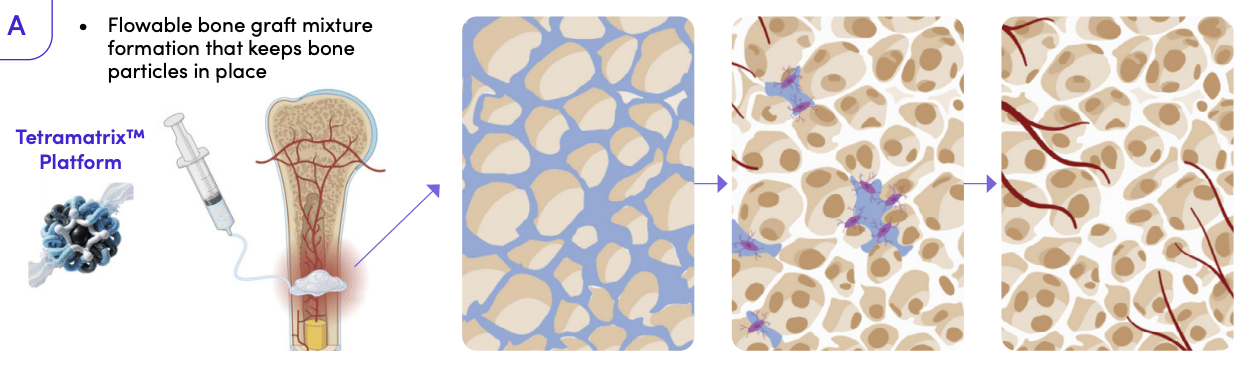
Bone Regeneration: Orthopedic
What’s the product?
Bone graft extender for orthopedic (knee) applications.
Who’s the distribution partner?
Yet to be selected, will be announced in 2026.
What stage is it at?
510(k) application in 2026.
What problem is it solving?
More and more orthopaedic surgeries are moving out of hospitals and into smaller day surgery clinics.
This shift is changing what products surgeons want to use.
In a big hospital, surgeons have large teams around them, plenty of time, and can work with complicated, multi-step bone repair products.
But in a day surgery clinic, the whole point is to get patients in and out quickly, so surgeons want products that are fast, simple, and ready to use straight out of the packet.
TTX’s orthopedic product fits this perfectly.
The second-generation product comes pre-filled in a syringe, requires no mixing or preparation, and is injected directly into the site. There's nothing to set up and nothing to waste.
A bone repair product built for day surgery, that’s the pitch…
Now it's about finding the right partner to deliver it.
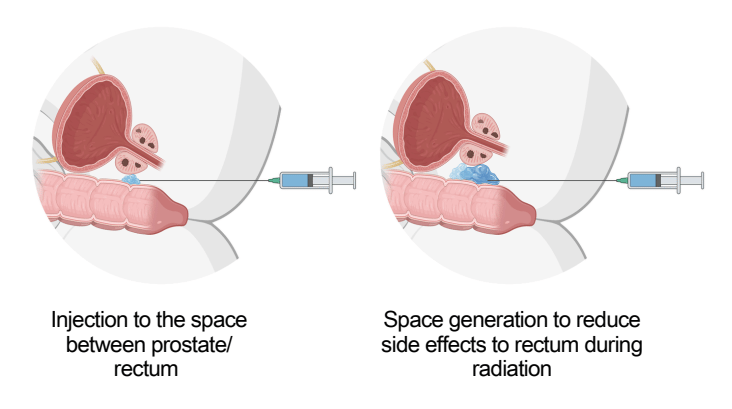
Spacer Technology: Cancers
What’s the product?
An injectable soft gel that hardens in the body, perfect for ‘spacing’ application in surgery (particularly oncology) to access different parts of the body.
Who’s the distribution partner?
None selected yet.
TTX has a JV partner to fund all clinical development separately, off its own balance sheet.
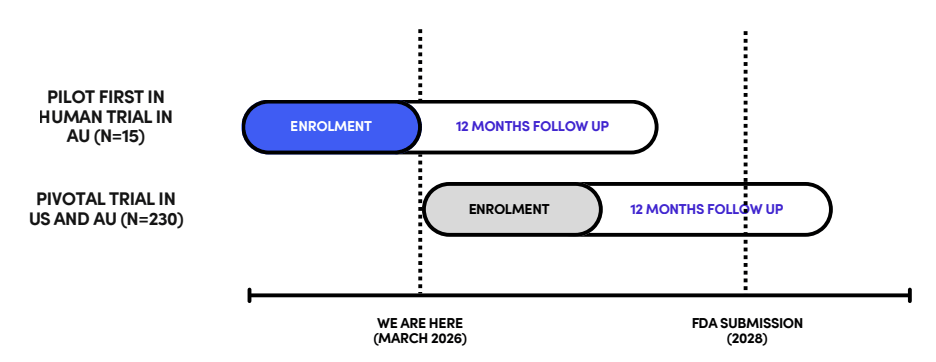
What stage is it at?
Enrolment complete for initial human pilot trial.
The product was considered safe, didn’t move, and was easy to handle.
Pivotal trial set to commence in 2026.
What problem is it solving?
There are two leaders in the space, SpaceOAR and Barrigel.
Both of these products were acquired by large pharma companies for ~US$600M (SpaceOAR in 2018 and Barrigel in 2023):

(Source, Fierce Biotech)

(Source, Fierce Biotech)
According to Surgeon Dan, who has used both of these products extensively:
SpaceOAR requires quite a steady hand, but if you put it in the wrong place, it can be really bad for the patient
Barrigel is much safer, but not as easy to handle, much more slippery, and doesn’t hold its shape as well.
They are both still good products, but each of them is flawed.
TTX is positioning its spacer technology at the intersection of the two: safe AND easy to handle.
A market with two entrenched players and no generic competition is exactly the kind of space where a differentiated third entrant like TTX can gain meaningful share.
Spacer Technology: Eyes
What’s the product?
Synthetic solution that maintains the volume and shape of the eye during surgeries like cataracts, and can be removed by a simple saline wash.
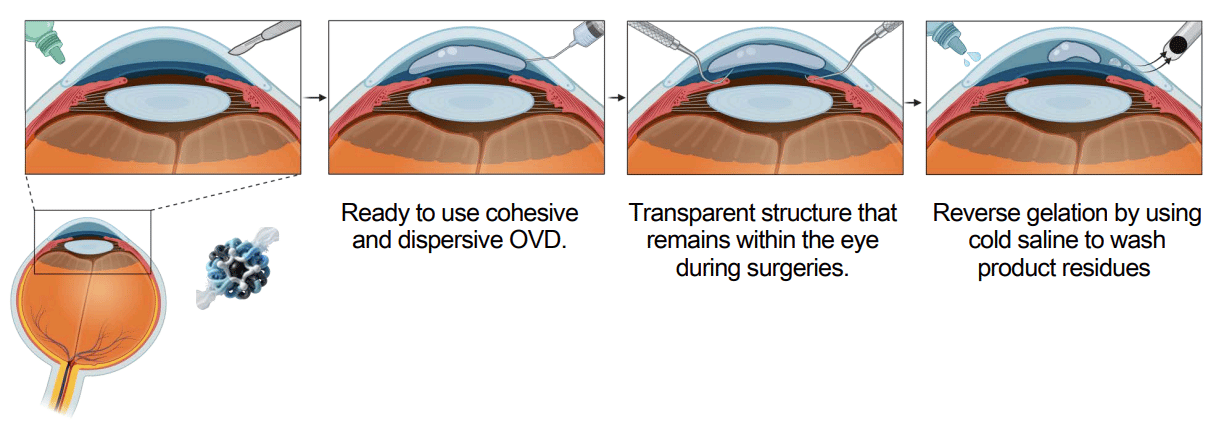
Who is the distribution partner?
TTX has signed a deal with Alcon, the market leader in this space, with a ~70% market share in eye spacer products.
What stage is it at?
Preclinical studies, funded by Alcon.
What’s the value proposition?
The ophthalmic spacer space is relatively well-established, with TTX’s partner, Alcon, holding around 70% market share.
The problem is not the product itself; it's the supply chain.
Alcon’s product is made from shark fins, which they get from one single supplier in China.
With tariffs and escalating tensions between the US and China, Alcon is desperately looking for an alternative product to diversify their supply chain risk.
The problem is that there hasn’t been much innovation in this space because, well, the products kind of work.
But for Alcon, finding an alternative like TTX (particularly one that is clear, stable, and easily removable) is crucial to solving their supply chain issues.
Scar Healing Technology
What’s the product?
A tissue healing product that can be used after each surgery to reduce (or potentially eliminate) scarring.
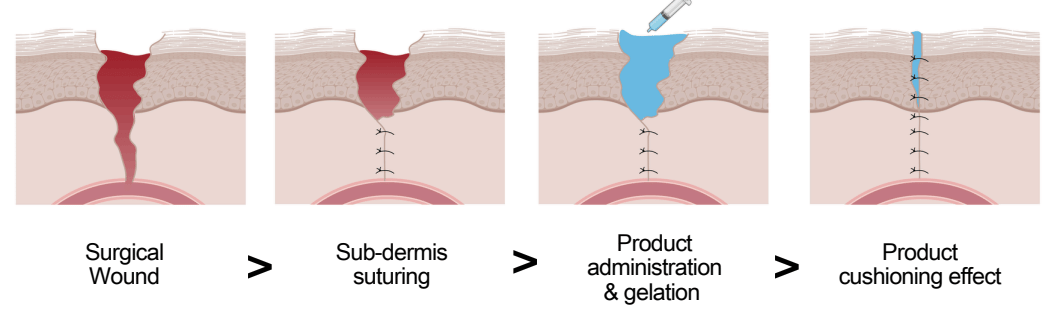
(If you ask Surgeon Dan, this was the product he was most excited about.)
Who is the distribution partner?
None, yet - too early.
What stage is it at?
Results have been published from the first cohort in the trial, with two other cohorts to go.
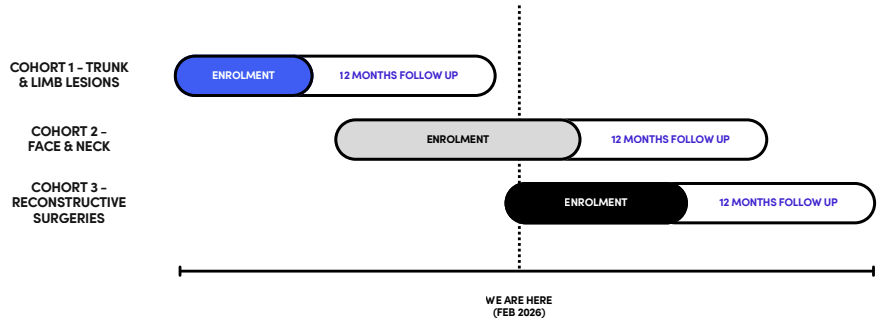
What’s the value proposition?
No scarring after surgery.
There is no product like this. It’s fully novel and in an untapped market.
The TAM is essentially every surgery.
Other products on the market can target healing after the scar has formed, but this product is the first to cause healing right at the time of incision.
So far, the results have been very good from early studies, with very low visible scaring 12-months post surgery:
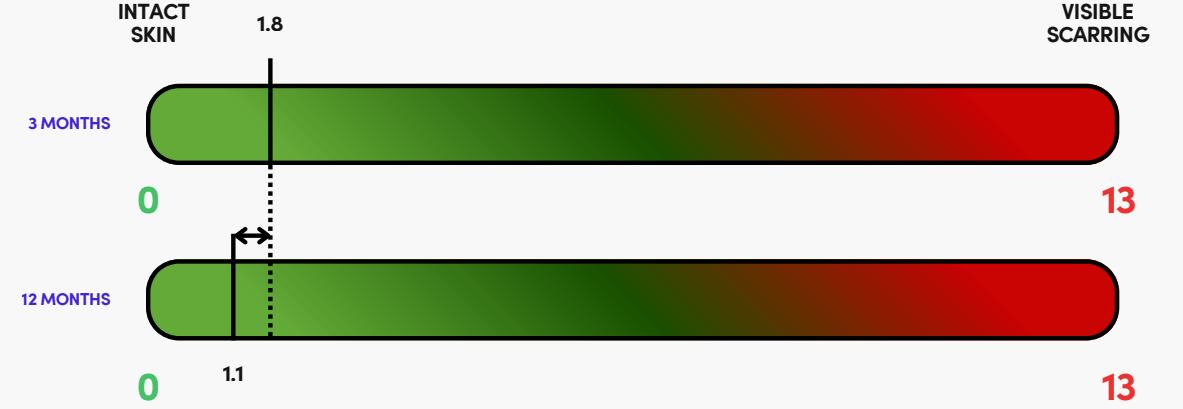
(Source, TTX Announcement)
This product, however, will need to follow the De Novo pathway for registration.
A long path to approval.
So it's a big blue-sky upside in TTX's portfolio, because there are genuinely no competitors out there.
What’s next for TTX?
(this one is for those that like to play the catlayts)
There are currently six products under development for TTX across different indications.
Two of them have decisions on FDA clearance this year.
FIRST, a bone-regeneration dental product, via the 510(k) pathway.
Application is in, decision pending.
SECOND, bone regeneration for orthopaedic (knee) surgeries.
TTX is still on the hunt for a go-to-market partner for this product.
Each of these catalysts gets TTX to the starting line to begin commercialising its product.
There are also study results expected this year from its skin scarring trials, and a pivotal trial is scheduled to commence for oncology spacing.
Here is the newsflow for TTX over the next 12 months:
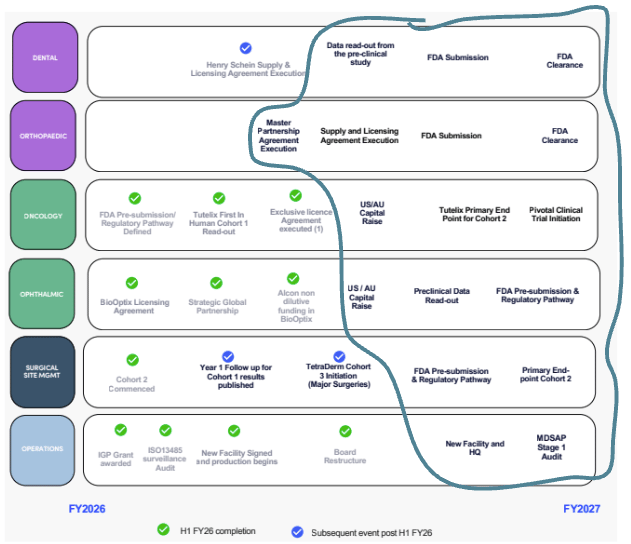
(Source, Half-Year Report, page 9)
So, there is a lot to look forward to…
The Risks
When evaluating any investment its important to pay attention to the risks.
Here are the ones that I’ve evaluated specifically for TTX…
Partnership/Commercial risk: TTX relies heavily on its partners. If TTX’s product is not well-integrated or partner reps have no incentive to sell, TTX’s sales are at risk.
Competition risk: TTX is selling a better version of an existing product. It will need to overcome the ‘switching cost’ for a surgeon to choose TTX over what they already use.
Regulatory risk: TTX has not yet received product approval. It will need to pass the FDA clearance check for each new indication that it targets.
Execution risk: Applying a SaaS business model to a biotech company is tricky. A lot will come down to execution and to managing multiple priorities at once.
Scale-up risk: TTX operates from a single lab in Sydney. To commercialise the product properly, it will need to invest in scaling up manufacturing production.
Market risk: If the world moves into a ‘risk-off’ environment, it could be bad for pre-revenue-generating companies like TTX.
The Armchair Take
I've been following TTX for about three months now, and everything they have done has impressed me.
The partner selection, the product design, the commercial discipline.
While the GLP-1 angle alone is enough to get my attention, TTX is so much more…
This is not a company throwing science at the wall and hoping something sticks; every product has a defined partner, a defined pathway, and a defined end user.
My first-ever Armchair Pick, and TTX earned it.
Big thank you to the CEO Will, for trusting me to tell the TTX story over the next 12 months.
If you’d like to read through an abridged version of this, here is my investment memo:
See you later today with the daily pulse check,
The Armchair Analyst
PS. If you have any questions about TTX, please reply. Provided there is enough interest, I’ll compile them all together and do a big ‘from the hip’ question and answer later in the week.