So, the next company on my Biotech 165 Challenge is very close to my heart.
My dad, who was a broker for 31 years, was heavily involved in this company.
A shareholder since the late 1990s.
A board member through a pretty tumultuous time in the company's history.
A cheerleader for this stock that could be on the cusp of a company making a Phase 3 result.
Of course, I’m talking about Avecho (ASX: AVE), which is just months away from an interim readout on a Phase 3 trial for insomnia.
He's watched the company go through its ups, downs, pivots, and false starts.
Multiple market cycles, more capital raises than I can count…
But Avecho always managed to reinvent itself.
Finally, after 25 years, this iteration might be the one that makes the company.
Avecho is just months away from an interim phase 3 clinical readout.
Its product?
A cannabis-based product for insomnia.
As I wrote in my CYP article, investing in biotech is sometimes more about timing the market than time in the market.
(Read the full article here: Playing the Catalysts: The Cynata (ASX: CYP) Story)
And the best thing to time around?
Data.
The more advanced in the clinical trial process, the greater the upside when results are good.
(But also the riskier the downside… as we saw last week with Immutep)
It’s a real punters market.
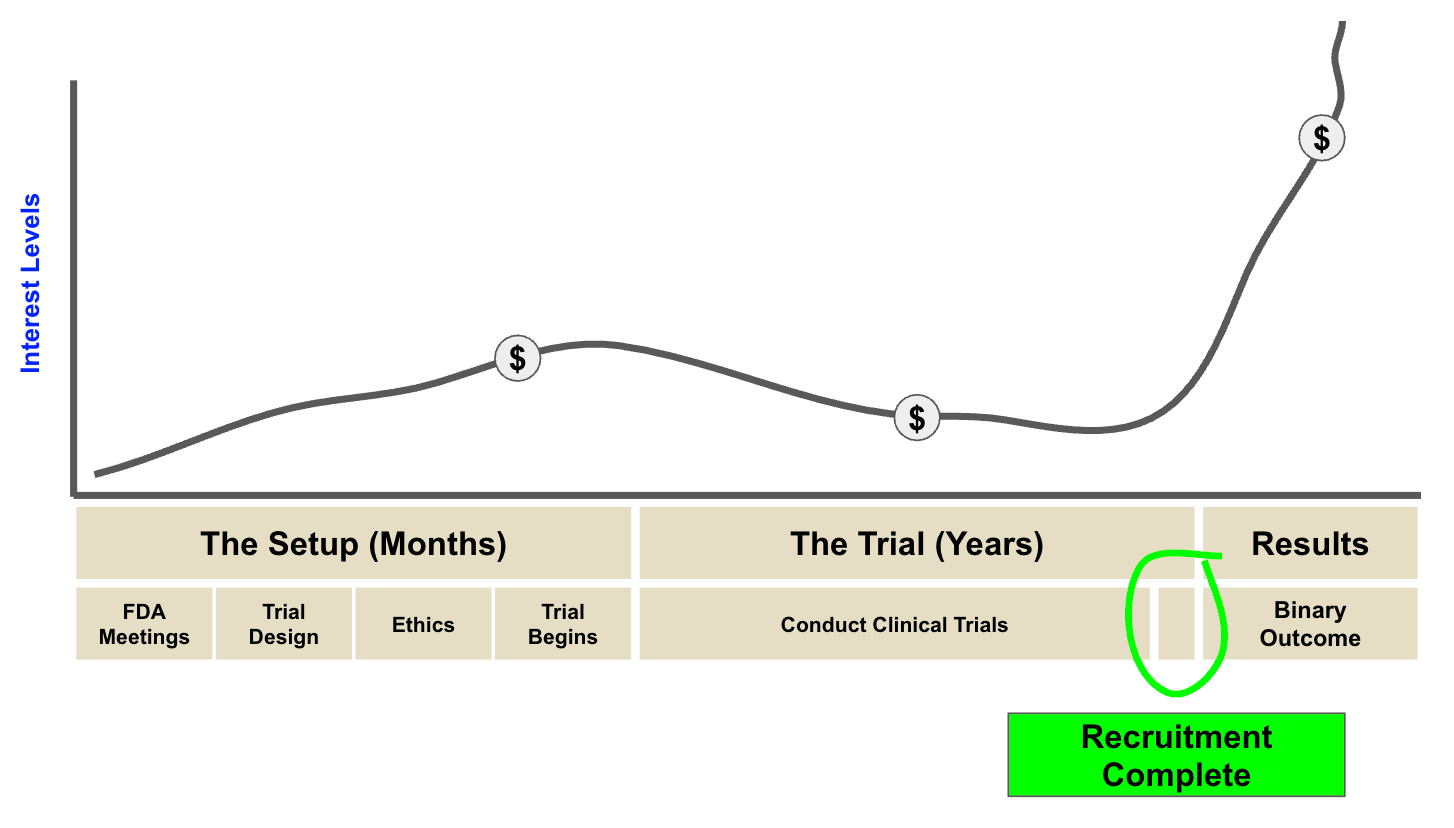
So, how do I like to play these catalysts?
This is a strategy I learned at my old firm:
Identify the catalyst early.
Invest when interest is low.
Top-slice in the lead-up to results as speculators enter.
And hold a decent portion through the readout itself.
The starting gun for investor interest is usually a "recruitment complete" announcement.
Because once recruitment is done, there's a clear timeframe for when results are coming.
No more delays.
Two weeks ago, Avecho announced it had completed recruitment for its Phase 3 study.
So there is a big catalyst on the horizon, but what is the upside if it pays off?
AVE will have the only cannabis based product that is sold over-the-counter in Australia.
Avecho has the backing of a major US$35 billion biopharma company Sandoz.
A company that knows the sleep market better than almost anyone.
The top-selling sleep product in Australia is Melatonin.
(holding around 40% market share)
Who sells Melatonin?
Sandoz.
If Avecho's product reaches the market, the commercial arrangement is already in place: Avecho collects 14–19% royalties on Sandoz's net sales.
And that's just Australia.
The company has already secured patents in Europe and the US, and I'd expect it to go after both markets in time.
There's one thing to keep an eye on, though: the listed options expire just before the interim readout.
These are exercisable at $0.012 (and the stock is trading around $0.010 and $0.012)
The results are now in the hands of the clinical trial gods.
So let's take a look at the story, the bet, and the risk vs reward for Avecho.
What's the story?
Avecho's core technology is TPM.
TPM is a patented drug-delivery platform that enhances drug absorption in the body.
(This has been around since the early days when Dad first invested).
The company has had a couple of cracks at developing a meaningful product from the TPM technology.
(skin creams, opioids, etc…)
But it was in April 2020, Avecho pivoted towards cannabis.
Medical cannabis was a natural fit for TPM because of one big problem with medical cannabis pills.
Bioavailability.
Bioavailability is the ability of the cannabis drug (and all its therapeutic effects) to be absorbed into the body through the gut.
Dose too low, not enough drug gets in the system.
Dose too high, it causes gastrointestinal issues.
So how do you deliver enough of the drug that doesn’t cause these gastrointestinal issues?
Avecho’s answer? TPM.
Shortly after the pivot to medical cannabis, the Australian Government down-scheduled cannabis, opening up two separate markets:
First, a medical cannabis market where doctors could prescribe cannabis flower, gummies, etc… (think Little Green Pharma, Cannatrek, Montu, Vita Life Sciences… a completely different playing field).
Second, and more interesting for Avecho: the possibility of an over-the-counter product.
The OTC market is enticing because it offers the lowest barrier to entry for anyone seeking CBD.
No prescription, just walk into a pharmacy and buy the goods.
But getting an OTC product approved hasn't been easy.
Those rules changed in 2021, and since then? Not a single approved product.
The ASX is littered with failed CBD clinical trials:
EcoFibre. Failed.
Cann Group. Failed.
BOD Science. Failed.
You can't blame the average punter for being sceptical about another ASX listed company going after OTC registration for a cannabis product.
But let's all say the line together:
"This time, it's different."

Well, I’m going to make the case that it actually could be.
How Avecho designed its trial to give itself the best chance of success
In 2020, the TGA changed the rules to allow companies to sell registered cannabis products over the counter, without a prescription, in sub-150mg doses.
Provided, of course, you could prove it was safe and effective in a clinical trial.
Four companies entered the race to be first.
Three have already failed.
Avecho is the last standing.
They failed for one reason - the placebo effect.
Insomnia is a neurological condition. If you think you're getting the medicine, you'll sleep better. This is the enemy of any sleep trial.
Avecho spent a significant amount of time before putting anyone in the trial screening out patients who responded to a placebo.
Remove the weeds before doing the gardening.
That's why the trial has taken so long.
Every patient recruited had to have an Insomnia Severity Index (ISI) score of 15, which is classified as clinical insomnia, not just a few bad nights of sleep.
For context: BOD Science's failed trial used an ISI score of 8, borderline on the sub-threshold for clinical insomnia.
By using stricter exclusion criteria, Avecho is reducing the risk of the placebo effect derailing its results and giving itself the best possible chance of success.
Will Avecho be the industry's “Steve Bradbury”?
If the trial outcomes are good, it will get the whole over-the-counter market all to itself.
What are the possible outcomes?
There are three scenarios:
Bear case: Interim results show the treatment isn't working. The trial stops. Avecho has to go back to the drawing board on what to do with TPM. This will be bad.
Bull case: Interim results indicate the treatment is working, but additional patient data is needed to complete registration. Avecho receives a US$16 million milestone payment from Sandoz. The trial continues with an additional 319 patients at more sites. Full results likely sometime in mid-to-late 2028.
Mega-bull case: The interim results are so strong that the trial can be stopped early and called complete. Avecho receives the US$16 million milestone payment from Sandoz. The company moves straight to registration, potentially putting a product on shelves far sooner than anyone expected.
Right now, the market is sceptical.
Which is fair.
Three trials have failed already.
But if Avecho succeeds, I'd expect a significant re-rate.
Particularly with non-dilutive milestone funding landing on the balance sheet.
The options dance
Avecho has a bunch of listed options exercisable at $0.012, currently trading near zero because of their short expiry date in May.
(Avecho is trading between $0.010 and $0.011 right now)
The market is attributing no value to them.
The question is: does the stock run before the options expire, putting the options into the money and creating a leveraged trade?
Or does the stock run after, clearing the overhang entirely and giving the share price a clear run?
A breakout through $0.012 will be challenging while those options are outstanding.
But once they expire, that ceiling goes away.
There's nothing the company can announce between now and then; it's all about how much investors want to punt on the prize.
With the Immutep disaster last week, I wouldn’t be surprised if punters are a little bit more skittish about binary trial results.
So we might not get the full ramp-up speculation that has happened in the past.
But it completely depends on risk appetite.
If the market is risk-on, expect a run.
If the market is risk-off, there might be a ceiling before results.
The size of the prize
There are really two markets here.
FIRST, Insomnia.
In Australia, there is effectively no approved OTC product for sleep.
Melatonin, sold by Sandoz, is one of the closest things to it, and in the US it's freely available in every pharmacy. But here, it's only prescribed to over-55s.
Every young professional who has ever dealt with sleep anxiety knows the trick of picking up melatonin on a US trip.
(My wife does it every time.)
There is a gaping hole in the Australian OTC sleep market.
Sandoz knows it, which is exactly why they backed Avecho.
Second: The broader CBD market.
While insomnia CBD products are what people will be buying on the shelves, the big commercial prize for the first mover to have an over-the-counter CBD-approved product.
This creates opportunities well beyond diagnosed insomniacs, particularly for those that are CBD self-medicated:
Anxiety-related sleep disruption, the most common co-morbidity with insomnia, is reachable through the same pharmacist conversation
Off-label pain and inflammation use, CBD has stronger clinical evidence for pain than melatonin
Grey market substitution, significant volumes of imported, unregulated CBD are already flowing into Australia; a legitimate OTC product captures those buyers
It’s not something that the company would ever talk about, but there is a portion of the market that will buy Avecho’s product for its ease of use.
The Armchair take
Speaking to CEO Paul Gavin, he shopped this product to big pharma companies everywhere.
The consistent response was: "It's just Australia. Too hard."
But not Sandoz.
Sandoz's CEO is the former Country Head for Australia and New Zealand, he knows the region.
And with Melatonin as a top-selling sleep product, they understand the commercial opportunity better than anyone.
Sometimes, these licensing deals are more about fit rather than the product alone.
Avecho got lucky that the right company, with the right muscle, came along at the right time.
It's now all about the results.
A big punt, and plenty have fallen over trying. But capped at ~$40 million, this is a relatively cheap option for a Phase 3 asset with major pharma backing.
(The only comparable I can remember was DXB with AdvanzPharma before those clinical results.)
The prize is clearly big. The trial design is the best attempt yet at cracking this market and Avecho is the last man standing with a very powerful partner in its corner.
But as we’ve seen with Immutep, there is no guarantee that the trial will work out as planned.
A big thank you to CEO Paul Gavin for walking me through the Avecho story.
See you all tomorrow,
The Armchair Analyst
The Pulse Check
Nyrada Inc. (ASX: NYR) is set to start its Phase IIa trial in April 2026 for myocardial ischemia reperfusion injury (heart attacks). Seven hospitals are participating, and 100 patients are targeted for enrolment. (NYR)
🪑 Good luck NYR shareholders. I’m looking forward to this one.
Nexalis Therapeutics (ASX: NX1) doses its first participant in its Phase 1 clinical trial of IRX-616a, a CBD-based inhalation aerosol for Panic Disorder. (NX1)
🪑 Short trial this one, final patient dosing is in June 2026. (Note, these guys are the old InhaleRX)
BlinkLab Limited (ASX: BB1) enters a trading halt pending the announcement of the launch of a nationwide autism screening program. (BB1)
🪑 This actually looks pretty interesting. 🍿
Little Green Pharma (ASX: LGP) terminates the proposed sale and leaseback of a production facility in Western Australia. (LGP)
🪑 Not ideal. Would have been an $7.8 million in non-dilutive funding for LGP that is now cancelled.
Immutep (ASX: IMM) completes the single ascending dose portion of its Phase I trial for autoimmune diseases. (IMM)
🪑 I get the continuous reporting obligations, but this is just a strange timing, and no mention of the Phase 3 study.
I would have liked to see the company do a presentation to shareholders BEFORE publishing news updates on other programs.
For transparency, I closed out my IMM trade yesterday at 5.5 cents. Tax loss!
Telix Pharmaseuticals (ASX: TLX) has been quietly rebounding from its low point in mid-February, up another ~5% yesterday.
🪑 A good chart discussion on Telix from Kneppy was published yesterday. Worth a watch.
Report: How AI is transforming radiology and diagnostic care (Healthy Innovations)
🪑 If you’re invested in AI detection companies like EchoIQ (ASX: EIQ), this report is worth a read.
A good high-level overview of AI in radiology (but can be equally applied to cardiology), including structural developments in the industry.
Cash Injection
Syntara (ASX: SNT) secures A$1.7M in a milestone payment from Parkinson’s UK, triggered by dosing the final patient in its Phase 2 trial for a specific REM sleep disorder that has been linked to Parkinson’s Disease. (SNT)
🪑 A big part of SNT’s strategy is to develop interesting technology and have others fund the development (kind of like a biotech incubator).
This is a good example of it.
SNT effectively gets a “free look” at the results of this trial funded by Parkinson’s UK.
EVE Health Group (ASX: EVE) upsizes its placement from $900K to $1.3M. (EVE)
🪑 Always good when the cornerstone investor tips more in.
Under the Microscope





